$3p_y$ orbital has which nodal plane?
A) XY
B) YZ
C) ZX
D) All of these.
Answer
581.4k+ views
Hint:First of all we should be aware of nodal planes. Hence, a nodal plane is a plane in which the probability of finding an electron is almost zero. The coordinates of these planes are obtained by solving the equation of Schrödinger wave for atoms or molecules such that to find the shape of atomic and molecular orbitals.
Complete step-by-step answer:There are only two nodes in a given orbital and these are Radial Node and the Angular Node.
The spherical surface where the probability of obtaining an electron is zero is termed as the Radial Node. Radial node is also known as the nodal region.
The term Angular node is also known as the nodal plane, the plane that passes through the nucleus. It is generally equal to the azimuthal quantum number (l).
Only the 2s orbital consists of a nodal shell, whereas the 2pz or the 2p orbitals have a nodal plane.
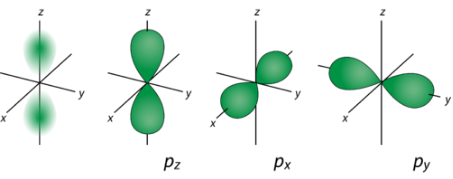
The formula for finding the total number of nodes is as follows.
Total no. of nodes = (n – l - 1).
Here, (n-1) is the total no. of nodes.
The option C has only value of (l=1).
Thus, $3p_y$ has ZX as the nodal plane.
Hence, the option C) is correct.
Note:The nodal surface is also known as a radial node. It is just a hollow spherical region where no electrons are found. There are three nodal planes found in 3p subshell, one from each (3px, 3py, 3pz).
Complete step-by-step answer:There are only two nodes in a given orbital and these are Radial Node and the Angular Node.
The spherical surface where the probability of obtaining an electron is zero is termed as the Radial Node. Radial node is also known as the nodal region.
The term Angular node is also known as the nodal plane, the plane that passes through the nucleus. It is generally equal to the azimuthal quantum number (l).
Only the 2s orbital consists of a nodal shell, whereas the 2pz or the 2p orbitals have a nodal plane.

The formula for finding the total number of nodes is as follows.
Total no. of nodes = (n – l - 1).
Here, (n-1) is the total no. of nodes.
The option C has only value of (l=1).
Thus, $3p_y$ has ZX as the nodal plane.
Hence, the option C) is correct.
Note:The nodal surface is also known as a radial node. It is just a hollow spherical region where no electrons are found. There are three nodal planes found in 3p subshell, one from each (3px, 3py, 3pz).
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Class 11 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Accountancy: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

In what year Guru Nanak Dev ji was born A15 April 1469 class 11 social science CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE




