1. Define standard enthalpy of combustion.
2. calculate the enthalpy change for the reaction:
${{N}_{2(g)}}+3{{H}_{2(g)}}\to 2N{{H}_{3(g)}}$
The bond enthalpies are:
Bond
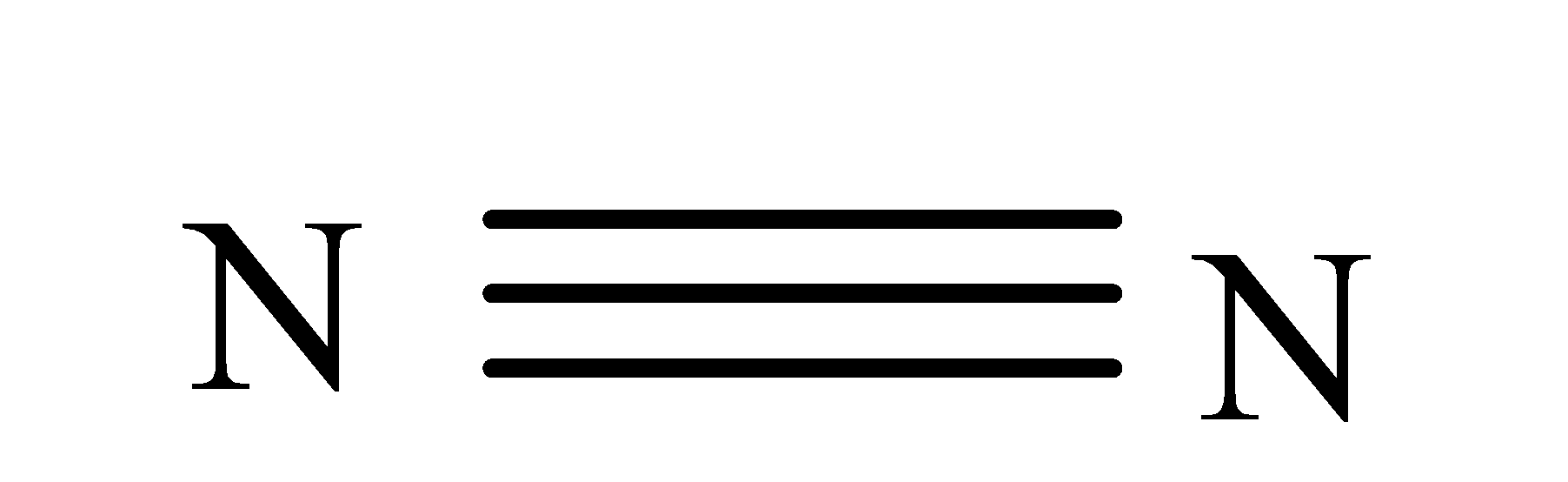 H-H N-H
H-H N-H
$\Delta {{H}^{\circ }}kJ mol{{e}^{-1}}\text{ }$ 946 435 389
Answer
602.1k+ views
Hint: By combustion we mean any substance which burns in the presence of excess of air and by standard state we mean the standard conditions of temperature and pressure i.e. 1 atm pressure and 273K temperature. And by the enthalpy change we means the amount of heat released or absorbed when pressure is constant and can be calculated by using the formula as; $\text{Enthalpy change }\Delta {{H}^{\circ }}\text{=}\sum \Delta \text{H( reactants)-}\sum \Delta \text{H( products)}$. Now answer the statement.
Complete answer:
1. By the term standard enthalpy of combustion we means the change produced in the enthalpy when one mole of the substance or any compound is burnt in the presence of excess of oxygen and all the reactants and products are present in their standard states i.e. in their gaseous states and under standard conditions of temperature and pressure i.e. 1 atm pressure and 273 K temperature. Example:
\[{{C}_{3}}{{H}_{8}}(g)+5{{O}_{2}}(g)\to 3C{{O}_{2}}(g0+4{{H}_{2}}O(g)\]
Now coming to the second part.
2.First of all. Let’s discuss what enthalpy changes . By the enthalpy change we means the amount of heat taken or given out in any reaction which occurs at the constant pressure and it denoted by the symbol as$\Delta {{H}^{\circ }}$and its symbols are $kJ mol{{e}^{-1}}\text{ }$.
The bond enthalpies of the reaction are as;
${{N}_{2(g)}}+3{{H}_{2(g)}}\to 2N{{H}_{3(g)}}$
Bond enthalpy of ammonia $\Delta {{H}^{\circ }}kJ mol{{e}^{-1}}\text{ }$= 389$kJmol{{e}^{-1}}\text{ }$
Bond enthalpy of Nitrogen $\Delta {{H}^{\circ }}kJ mol{{e}^{-1}}\text{ }$= 946$kJmol{{e}^{-1}}\text{ }$
Bond enthalpy of hydrogen $\Delta {{H}^{\circ }}kJ mol{{e}^{-1}}\text{ }$= 435$kJmol{{e}^{-1}}\text{ }$
So, now the enthalpy change for the reaction can be calculated as;
$\begin{align}
& \text{Enthalpy change }\Delta {{H}^{\circ }}\text{=}\sum \Delta \text{H( reactants)-}\sum \Delta \text{H( products)} \\
& \text{ =}\sum \Delta {{\text{H}}_{{{N}_{2}}}}+\sum 3\Delta {{\text{H}}_{{{H}_{2}}}}-\sum 6\Delta {{\text{H}}_{N{{H}_{3}}}} \\
& \text{ = 946+3}\times \text{435-6}\times 389 \\
& \text{ = 946+ 1305-2334} \\
& \text{ =- 83 kJ mol}{{\text{e}}^{-1}} \\
\end{align}$
Therefore, the enthalpy changes $\Delta {{H}^{\circ }}$ for the reaction is -83$kJmol{{e}^{-1}}\text{ }$
Note:
To calculate the enthalpy change always takes the units in their standard states and under standard conditions of temperature and pressure and if the substances are present in the solid state, then their enthalpy is always taken as zero.
Complete answer:
1. By the term standard enthalpy of combustion we means the change produced in the enthalpy when one mole of the substance or any compound is burnt in the presence of excess of oxygen and all the reactants and products are present in their standard states i.e. in their gaseous states and under standard conditions of temperature and pressure i.e. 1 atm pressure and 273 K temperature. Example:
\[{{C}_{3}}{{H}_{8}}(g)+5{{O}_{2}}(g)\to 3C{{O}_{2}}(g0+4{{H}_{2}}O(g)\]
Now coming to the second part.
2.First of all. Let’s discuss what enthalpy changes . By the enthalpy change we means the amount of heat taken or given out in any reaction which occurs at the constant pressure and it denoted by the symbol as$\Delta {{H}^{\circ }}$and its symbols are $kJ mol{{e}^{-1}}\text{ }$.
The bond enthalpies of the reaction are as;
${{N}_{2(g)}}+3{{H}_{2(g)}}\to 2N{{H}_{3(g)}}$
Bond enthalpy of ammonia $\Delta {{H}^{\circ }}kJ mol{{e}^{-1}}\text{ }$= 389$kJmol{{e}^{-1}}\text{ }$
Bond enthalpy of Nitrogen $\Delta {{H}^{\circ }}kJ mol{{e}^{-1}}\text{ }$= 946$kJmol{{e}^{-1}}\text{ }$
Bond enthalpy of hydrogen $\Delta {{H}^{\circ }}kJ mol{{e}^{-1}}\text{ }$= 435$kJmol{{e}^{-1}}\text{ }$
So, now the enthalpy change for the reaction can be calculated as;
$\begin{align}
& \text{Enthalpy change }\Delta {{H}^{\circ }}\text{=}\sum \Delta \text{H( reactants)-}\sum \Delta \text{H( products)} \\
& \text{ =}\sum \Delta {{\text{H}}_{{{N}_{2}}}}+\sum 3\Delta {{\text{H}}_{{{H}_{2}}}}-\sum 6\Delta {{\text{H}}_{N{{H}_{3}}}} \\
& \text{ = 946+3}\times \text{435-6}\times 389 \\
& \text{ = 946+ 1305-2334} \\
& \text{ =- 83 kJ mol}{{\text{e}}^{-1}} \\
\end{align}$
Therefore, the enthalpy changes $\Delta {{H}^{\circ }}$ for the reaction is -83$kJmol{{e}^{-1}}\text{ }$
Note:
To calculate the enthalpy change always takes the units in their standard states and under standard conditions of temperature and pressure and if the substances are present in the solid state, then their enthalpy is always taken as zero.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




