Write the structure of orthophosphorous acid and give its basicity.
Answer
616.8k+ views
Hint
Basicity is determined by the number of replaceable hydrogen atoms. If an acid contains 1 replaceable hydrogen, then its basicity is equal to 1 and known as monobasic. If an acid contains 2 replaceable hydrogen atoms, then its basicity is 2 and known as dibasic. And if an acid contains 3 replaceable hydrogen atoms then its basicity is 3 and known as tribasic.
Complete step by step solution
Orthophosphorous acid is also called phosphorous acid and has the formula ${{\rm{H}}_{\rm{3}}}{\rm{P}}{{\rm{O}}_{\rm{3}}}$. It is also a mineral acid.
It is generally colourless and odourless. It is soluble in water and it forms a white crystals.it acts as a strong reducing agent and it can easily reduce many compounds like mercuric chloride to mercurous chloride, copper sulphate to metallic copper, silver nitrate to metallic silver, iodine to hydriodic acid etc.
Orthophosphorous acid when heated decomposes to give phosphoric acid and phosphine. It is prepared by the reaction of phosphorous trichloride and cold water. It is also prepared by the reaction of phosphorus trichloride and anhydrous oxalic acid.
The structure of Orthophosphorous acid is given below
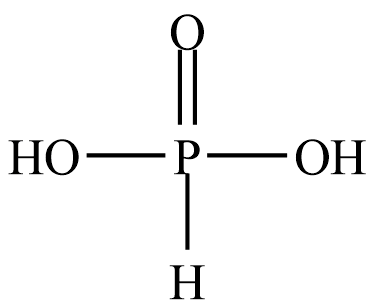
It can be seen that there are two $ - {\rm{OH}}$ groups present. There is a large electronegativity difference between the oxygen and hydrogen atoms. Due to which these two hydrogen atoms can easily remove. So, there are two numbers of replaceable hydrogen atoms.
Hence, its basicity is two.
Note:
Orthophosphorous acid is widely used in different industries namely the agricultural and pharmaceutical sector. It is used in the removal of rusting. It is also widely used in the food industry. It also helps to maintain the level of acid in food like meats, cheese etc. It is also used in various beauty products like dyes, nail products, make up etc.
Basicity is determined by the number of replaceable hydrogen atoms. If an acid contains 1 replaceable hydrogen, then its basicity is equal to 1 and known as monobasic. If an acid contains 2 replaceable hydrogen atoms, then its basicity is 2 and known as dibasic. And if an acid contains 3 replaceable hydrogen atoms then its basicity is 3 and known as tribasic.
Complete step by step solution
Orthophosphorous acid is also called phosphorous acid and has the formula ${{\rm{H}}_{\rm{3}}}{\rm{P}}{{\rm{O}}_{\rm{3}}}$. It is also a mineral acid.
It is generally colourless and odourless. It is soluble in water and it forms a white crystals.it acts as a strong reducing agent and it can easily reduce many compounds like mercuric chloride to mercurous chloride, copper sulphate to metallic copper, silver nitrate to metallic silver, iodine to hydriodic acid etc.
Orthophosphorous acid when heated decomposes to give phosphoric acid and phosphine. It is prepared by the reaction of phosphorous trichloride and cold water. It is also prepared by the reaction of phosphorus trichloride and anhydrous oxalic acid.
The structure of Orthophosphorous acid is given below

It can be seen that there are two $ - {\rm{OH}}$ groups present. There is a large electronegativity difference between the oxygen and hydrogen atoms. Due to which these two hydrogen atoms can easily remove. So, there are two numbers of replaceable hydrogen atoms.
Hence, its basicity is two.
Note:
Orthophosphorous acid is widely used in different industries namely the agricultural and pharmaceutical sector. It is used in the removal of rusting. It is also widely used in the food industry. It also helps to maintain the level of acid in food like meats, cheese etc. It is also used in various beauty products like dyes, nail products, make up etc.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Explain the Treaty of Vienna of 1815 class 10 social science CBSE

Why is it 530 pm in india when it is 1200 afternoon class 10 social science CBSE

What is the full form of POSCO class 10 social science CBSE

Define Potential, Developed, Stock and Reserved resources

Which Country Has the Largest Border with India?

Complete the sentence with the most appropriate word class 10 english CBSE




