Write the lewis structure of Oxygen (${{O}_{2}}$) and ethyne (${{C}_{2}}{{H}_{2}}$) molecules.
Answer
596.4k+ views
Hint: A Lewis Structure is a very simplified representation of the valence shell electrons in a molecule. It is used to show how the electrons are arranged around individual atoms in a molecule. Electrons are shown as "dots" or for bonding electrons as a line between the two atoms. The goal is to obtain the "best" electron configuration, i.e. the octet rule and formal charges need to be satisfied.
Complete step by step answer:
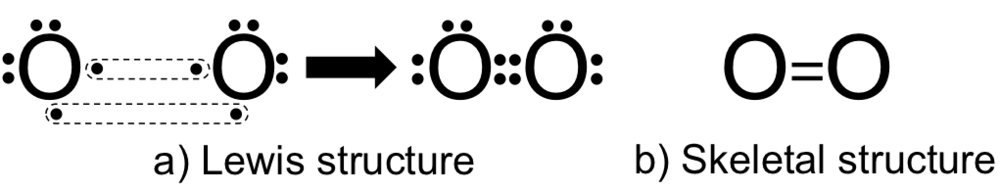
Electronic configuration of oxygen atom: \[1{{s}^{2}}2{{s}^{2}}2{{p}^{4}}\]
Oxygen atom has six electrons in its valence shell and to fulfill its octet it needs 2 more electrons.
- Now here for these two electrons it is combined with another oxygen atom and both atoms will 2 electrons a piece and make a double bond between them, so four electrons will be shared.
Lewis structure is shown below:
Lewis structure of ethyne:
- Electronic configuration of Carbon atom: \[1{{s}^{2}}2{{s}^{2}}2{{p}^{2}}\]
To show tetravalency one 2s electron will move into empty 2p orbital
New electronic configuration of Carbon atom: \[1{{s}^{2}}2{{s}^{1}}2{{p}^{3}}\]
To complete its octet each carbon needs 4 electrons.
Electronic configuration of hydrogen: \[1{{s}^{1}}\]
To have a configuration like He, hydrogen needs one electron.
- So here both carbons can combine with one hydrogen but that will only give each carbon one electron and for the remaining three electrons, these carbons will combine together and share 3 electrons a piece and a triple bond will form and now each has 8 electrons, where 1 electron it is getting from 1 hydrogen and three electrons it is getting from other carbon atom.
Its lewis structure is shown below:
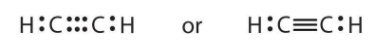
Additional Information :Total number of bonds between two atoms will always be equal to half of the number of total electrons shared. As in above case 6 electrons were shared that’s why both carbon atoms have triple bonds between them.
Note: Ethyne is a colorless gas with a characteristic smell. It has a melting point $-81{}^{0}C$. It has a boiling point $-84{}^{0}C$. It is a poisonous gas and liquid ethyne is explosive. It is lighter than air and significantly soluble in water. It has linear structure.
Complete step by step answer:
Electronic configuration of oxygen atom: \[1{{s}^{2}}2{{s}^{2}}2{{p}^{4}}\]
Oxygen atom has six electrons in its valence shell and to fulfill its octet it needs 2 more electrons.
- Now here for these two electrons it is combined with another oxygen atom and both atoms will 2 electrons a piece and make a double bond between them, so four electrons will be shared.
Lewis structure is shown below:

Lewis structure of ethyne:
- Electronic configuration of Carbon atom: \[1{{s}^{2}}2{{s}^{2}}2{{p}^{2}}\]
To show tetravalency one 2s electron will move into empty 2p orbital
New electronic configuration of Carbon atom: \[1{{s}^{2}}2{{s}^{1}}2{{p}^{3}}\]
To complete its octet each carbon needs 4 electrons.
Electronic configuration of hydrogen: \[1{{s}^{1}}\]
To have a configuration like He, hydrogen needs one electron.
- So here both carbons can combine with one hydrogen but that will only give each carbon one electron and for the remaining three electrons, these carbons will combine together and share 3 electrons a piece and a triple bond will form and now each has 8 electrons, where 1 electron it is getting from 1 hydrogen and three electrons it is getting from other carbon atom.
Its lewis structure is shown below:
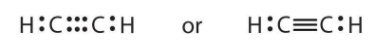
Additional Information :Total number of bonds between two atoms will always be equal to half of the number of total electrons shared. As in above case 6 electrons were shared that’s why both carbon atoms have triple bonds between them.
Note: Ethyne is a colorless gas with a characteristic smell. It has a melting point $-81{}^{0}C$. It has a boiling point $-84{}^{0}C$. It is a poisonous gas and liquid ethyne is explosive. It is lighter than air and significantly soluble in water. It has linear structure.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

What are hard magnetic materials class 11 physics CBSE




