Write down the rules of IUPAC Nomenclature.
Answer
620.4k+ views
Hint: The IUPAC devised a set of instructions or rules that helped in naming the organic molecules. This helped scientists to derive names of the molecules which were not even discovered. These rules have been revised from time to time to improve the quality of nomenclature.
Complete answer :
The IUPAC stands for International Union of Pure and Applied Chemistry. It has formulated some rules for the systematic nomenclature of organic compounds which are even revised later on for the uniform naming of compounds. The IUPAC has laid down some rules which govern the naming of all the organic compounds. This is necessary so that there is uniform nomenclature to be followed world-wide.
The IUPAC name of any molecule consists of three parts-
Prefix, stem name and suffix.
The rules for nomenclature are -
Rule 1 :- The longest chain in the molecule will be the parent chain. So, the first thing one should do is to find the longest carbon chain in the molecule.
Example - In the following molecule, the correct numbering is as-
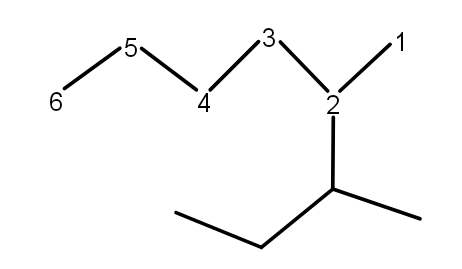
The molecule has the longest chain of six carbon atoms.
Rule 2 :- Find out all the substituents present on the carbon chain. It can be any- from methyl to any alcohol substituent.
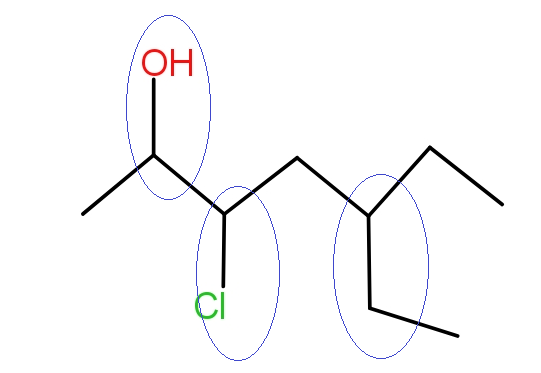
Rule 3 :- Start numbering the parent chain from the end such that the substituent gets the lowest number. This is called the lowest locant rule.
For example - Numbering in this chain will be as -
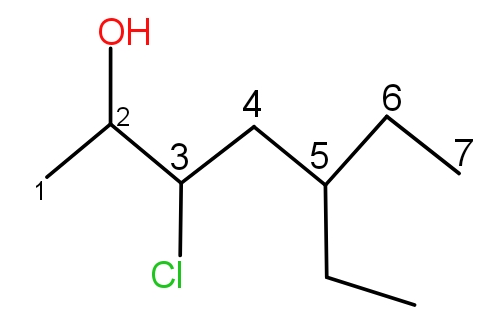
Rule 4 :- If the substituent occurs more than one time, then the prefix di, tri or tetra is being used with the location of the carbon on which it is present.
Example -
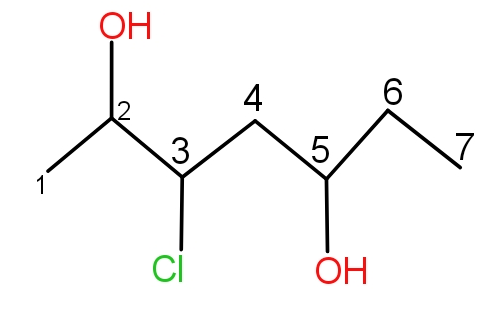
This has IUPAC name as 3 chloro-hept-2,5-diol.
Rule 5 :- If more than one substituents are present and they are different. So, these will be named in alphabetical order.
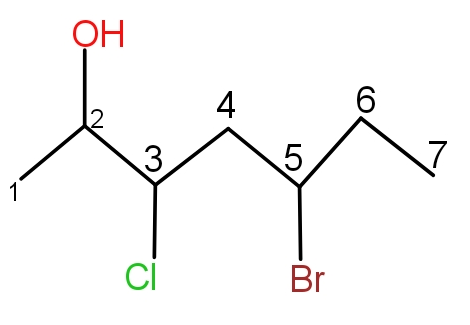
During naming of this molecule, bromine will come first and then chlorine. Thus, the name is 5-bromo,-3chlorohept-2-ol.
Rule 6 :- A cyclic hydrocarbon has a prefix cyclo before the name.
Example - The following molecules have names as-
Note :
The IUPAC nomenclature is needed because earlier every chemist gave its own new name to the molecule it discovered. As a result, one molecule had different names at different places. It was difficult to study them. No one got to know whether the molecule discovered by him/her is the new one or the one that has already been discovered. So, the scientist devised some formulations to uniform the naming of compounds.
Complete answer :
The IUPAC stands for International Union of Pure and Applied Chemistry. It has formulated some rules for the systematic nomenclature of organic compounds which are even revised later on for the uniform naming of compounds. The IUPAC has laid down some rules which govern the naming of all the organic compounds. This is necessary so that there is uniform nomenclature to be followed world-wide.
The IUPAC name of any molecule consists of three parts-
Prefix, stem name and suffix.
The rules for nomenclature are -
Rule 1 :- The longest chain in the molecule will be the parent chain. So, the first thing one should do is to find the longest carbon chain in the molecule.
Example - In the following molecule, the correct numbering is as-

The molecule has the longest chain of six carbon atoms.
Rule 2 :- Find out all the substituents present on the carbon chain. It can be any- from methyl to any alcohol substituent.

Rule 3 :- Start numbering the parent chain from the end such that the substituent gets the lowest number. This is called the lowest locant rule.
For example - Numbering in this chain will be as -

Rule 4 :- If the substituent occurs more than one time, then the prefix di, tri or tetra is being used with the location of the carbon on which it is present.
Example -

This has IUPAC name as 3 chloro-hept-2,5-diol.
Rule 5 :- If more than one substituents are present and they are different. So, these will be named in alphabetical order.

During naming of this molecule, bromine will come first and then chlorine. Thus, the name is 5-bromo,-3chlorohept-2-ol.
Rule 6 :- A cyclic hydrocarbon has a prefix cyclo before the name.
Example - The following molecules have names as-


Note :
The IUPAC nomenclature is needed because earlier every chemist gave its own new name to the molecule it discovered. As a result, one molecule had different names at different places. It was difficult to study them. No one got to know whether the molecule discovered by him/her is the new one or the one that has already been discovered. So, the scientist devised some formulations to uniform the naming of compounds.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE




