Which oxyacid of sulphur contains S-S single bond?
A.Oleum
B.Marshall’s acid
C.Dithionic acid
D.Thiosulphuric acid
Answer
598.5k+ views
Hint: In oxyacid of sulphur, sulphur is the central atom exhibiting a tetrahedral structure when coordinated with oxygen. This contains at least one \[S = O\] bond and one \[S - OH\] bond. In addition to these, a chain of \[{( - S - )_n}\] as in \[{H_2}{S_2}{O_6}\] . Such oxyacids with \[S - S\] linkages are called Thioacids.
Complete step-by-step solution:
The sulphur oxyacids are the acids that contain oxygen, hydrogen and sulphur. These are divides into four groups based on their structural similarities:
Sulphurous acid group- these are prepared by dissolving sulphur dioxide in water. Their structure is pyramidal with three oxygen atoms on a triangle i.e. tetrahedral structure is distorted by lone pairs. These are strong reducing agents and have bleaching properties.
For example, sulphurous acid \[{H_2}S{O_3}\] .
Sulphuric acid group- these are also called oil of vitriol, produced by lead chamber and contact processes by dissolving \[S{O_3}\] in water. For example, sulphuric acid \[{H_2}S{O_4}\] (oleum) and thiosulphuric acid.
Thionic acid group- these are a series of unstable acids with general formula of \[{H_2}{S_n}{O_6}\] where \[n = 2\] to 6. For example, Dithionic acid \[{H_2}{S_2}{O_6}\] .
Peroxo acid group- these are also called Marshall’s acid or Caro’s acid. The central atom sulphur has +6 oxidation state with a peroxo group in it. These are derived from hydrogen peroxide by replacing hydrogen atoms. For example, peroxydisulfuric acid \[{H_2}{S_2}{O_8}\] .
From all these series, we saw that the \[S - S\] bond is present only in thionic acid series. For example, dithionic acid \[{H_2}{S_2}{O_6}\] .
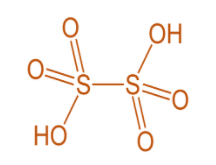
Hence, the correct option is (C).
Note: All oxyacids have the acidic hydrogen bonded to an oxygen atom. i.e. \[ - OH\] bond. The electronegativity of the central atom and the number of O atoms are responsible for the oxyacid acidity.
Complete step-by-step solution:
The sulphur oxyacids are the acids that contain oxygen, hydrogen and sulphur. These are divides into four groups based on their structural similarities:
Sulphurous acid group- these are prepared by dissolving sulphur dioxide in water. Their structure is pyramidal with three oxygen atoms on a triangle i.e. tetrahedral structure is distorted by lone pairs. These are strong reducing agents and have bleaching properties.
For example, sulphurous acid \[{H_2}S{O_3}\] .
Sulphuric acid group- these are also called oil of vitriol, produced by lead chamber and contact processes by dissolving \[S{O_3}\] in water. For example, sulphuric acid \[{H_2}S{O_4}\] (oleum) and thiosulphuric acid.
Thionic acid group- these are a series of unstable acids with general formula of \[{H_2}{S_n}{O_6}\] where \[n = 2\] to 6. For example, Dithionic acid \[{H_2}{S_2}{O_6}\] .
Peroxo acid group- these are also called Marshall’s acid or Caro’s acid. The central atom sulphur has +6 oxidation state with a peroxo group in it. These are derived from hydrogen peroxide by replacing hydrogen atoms. For example, peroxydisulfuric acid \[{H_2}{S_2}{O_8}\] .
From all these series, we saw that the \[S - S\] bond is present only in thionic acid series. For example, dithionic acid \[{H_2}{S_2}{O_6}\] .

Hence, the correct option is (C).
Note: All oxyacids have the acidic hydrogen bonded to an oxygen atom. i.e. \[ - OH\] bond. The electronegativity of the central atom and the number of O atoms are responsible for the oxyacid acidity.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Which among the following are examples of coming together class 11 social science CBSE




