Which of the following will be soluble in sodium bicarbonate solution?
A.
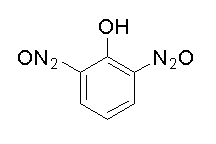
B.
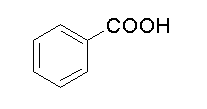
C.
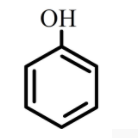
D.
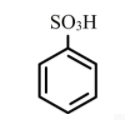




Answer
584.4k+ views
Hint: Solubility of the acid component and base component is dependent on the strength of the acid and base. Sodium bicarbonate is a weak base which reacts with strong organic acids which have pKa range from 4 to 5 liberated carbon dioxide gas.
Complete step by step answer:
The given compound sodium bicarbonate solution is a weak base, it reacts with strong acid to form salt and water by releasing carbon dioxide.
The solubility of strong acid is high in weak base as compared to the solubility of weak acid in weak base.
The given compound 2,6 dinitrophenol is a strong acid, it will dissolve in sodium bicarbonate to form salt and water by liberating carbon dioxide.
The benzene sulphonic acid is a strong acid, therefore it will dissolve in sodium bicarbonate solution to form salt and water by liberating carbon dioxide.
The benzoic acid is also a strong acid, therefore it will dissolve in sodium bicarbonate solution to form salt and water by liberating carbon dioxide..
The compound phenol is considered as a weak acid. Therefore, it will not dissolve in sodium bicarbonate solution and does not release carbon dioxide.
Therefore, the correct option is A, B, and D.
Note:
$NaHC{O_3}$ contains $N{a^ + }$ and $HC{O_3}^ -$ ion. The pKa range of phenol is between 8 to 14 so the bicarbonate is not able to deprotonate the phenol functional group and thus being an acid also phenol does not give any reaction.
Complete step by step answer:
The given compound sodium bicarbonate solution is a weak base, it reacts with strong acid to form salt and water by releasing carbon dioxide.
The solubility of strong acid is high in weak base as compared to the solubility of weak acid in weak base.
The given compound 2,6 dinitrophenol is a strong acid, it will dissolve in sodium bicarbonate to form salt and water by liberating carbon dioxide.
The benzene sulphonic acid is a strong acid, therefore it will dissolve in sodium bicarbonate solution to form salt and water by liberating carbon dioxide.
The benzoic acid is also a strong acid, therefore it will dissolve in sodium bicarbonate solution to form salt and water by liberating carbon dioxide..
The compound phenol is considered as a weak acid. Therefore, it will not dissolve in sodium bicarbonate solution and does not release carbon dioxide.
Therefore, the correct option is A, B, and D.
Note:
$NaHC{O_3}$ contains $N{a^ + }$ and $HC{O_3}^ -$ ion. The pKa range of phenol is between 8 to 14 so the bicarbonate is not able to deprotonate the phenol functional group and thus being an acid also phenol does not give any reaction.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




