Which of the following pairs of d-orbitals will have electron density along the axis.
A. ${{\text{d}}_{{{\text{z}}^{\text{2}}}}}{\text{,}}{{\text{d}}_{{{\text{x}}^2} - {{\text{y}}^{\text{2}}}}}$
B. ${{\text{d}}_{{\text{xy}}}},{{\text{d}}_{{{\text{x}}^2} - {{\text{y}}^{\text{2}}}}}$
C. ${{\text{d}}_{{{\text{z}}^{\text{2}}}}}{\text{,}}\,{{\text{d}}_{xz}}$
D. ${{\text{d}}_{xz}},{{\text{d}}_{yz}}$
Answer
594.3k+ views
Hint: The ${{\text{d}}_{{{\text{z}}^{\text{2}}}}}$ and ${{\text{d}}_{{{\text{x}}^2} - {{\text{y}}^{\text{2}}}}}$ orbitals which lies on the axis and the ${{\text{d}}_{xz}}$,${{\text{d}}_{yz}}$, and ${{\text{d}}_{xy}}$ lies in between the axis. Total five orbitals are present in the d - orbitals.
Complete step by step answer:
The position of electron density depends upon the position of the orbitals.
The d-orbital is a set of five denigrate orbitals named as ${{\text{d}}_{{{\text{z}}^{\text{2}}}}}$, ${{\text{d}}_{{{\text{x}}^2} - {{\text{y}}^{\text{2}}}}}$, ${{\text{d}}_{xz}}$,${{\text{d}}_{yz}}$, and ${{\text{d}}_{xy}}$.
The positions of these five d-orbitals on the axis is represented as follows:
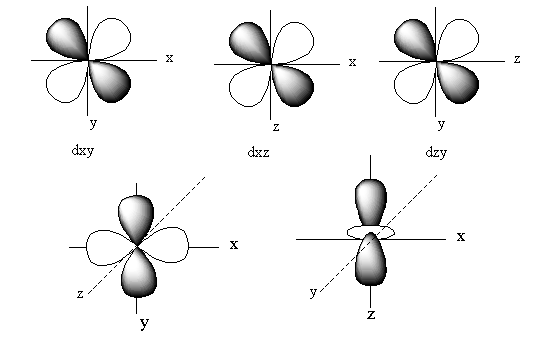
The orbitals of ${{\text{d}}_{xy}}$ lie in between the x and y-axis. So, the electron density of the ${{\text{d}}_{xy}}$ lies in between the x and y-axis.
The orbitals of ${{\text{d}}_{yz}}$ lie in between the z and y-axis. So, the electron density of the ${{\text{d}}_{yz}}$ lies in between the z and y-axis.
The orbitals of ${{\text{d}}_{xz}}$ lie in between the x and z-axis. So, the electron density of the ${{\text{d}}_{xz}}$ lies in between the x and z-axis.
The orbital of ${{\text{d}}_{{{\text{z}}^{\text{2}}}}}$lies on the z-axis. So, the electron density of the ${{\text{d}}_{{{\text{z}}^{\text{2}}}}}$lies on the z-axis.
The orbitals of ${{\text{d}}_{{{\text{x}}^2} - {{\text{y}}^{\text{2}}}}}$ lie on the x and y-axis. So, the electron density of the ${{\text{d}}_{{{\text{x}}^2} - {{\text{y}}^{\text{2}}}}}$ lies on the x and y-axis.
So, ${{\text{d}}_{{{\text{z}}^{\text{2}}}}}{\text{,}}{{\text{d}}_{{{\text{x}}^2} - {{\text{y}}^{\text{2}}}}}$ pairs of d-orbitals will have electron density along the axis.
Therefore, option (A) ${{\text{d}}_{{{\text{z}}^{\text{2}}}}}{\text{,}}{{\text{d}}_{{{\text{x}}^2} - {{\text{y}}^{\text{2}}}}}$ is correct.
Note: Out of five d-orbitals, two lie on the axis and three lie in between the axis. So, when ligands form a complex with the metal it affects the three orbitals differently and two orbitals differently, so the five d-orbitals break into two parts of three and two.
Complete step by step answer:
The position of electron density depends upon the position of the orbitals.
The d-orbital is a set of five denigrate orbitals named as ${{\text{d}}_{{{\text{z}}^{\text{2}}}}}$, ${{\text{d}}_{{{\text{x}}^2} - {{\text{y}}^{\text{2}}}}}$, ${{\text{d}}_{xz}}$,${{\text{d}}_{yz}}$, and ${{\text{d}}_{xy}}$.
The positions of these five d-orbitals on the axis is represented as follows:

The orbitals of ${{\text{d}}_{xy}}$ lie in between the x and y-axis. So, the electron density of the ${{\text{d}}_{xy}}$ lies in between the x and y-axis.
The orbitals of ${{\text{d}}_{yz}}$ lie in between the z and y-axis. So, the electron density of the ${{\text{d}}_{yz}}$ lies in between the z and y-axis.
The orbitals of ${{\text{d}}_{xz}}$ lie in between the x and z-axis. So, the electron density of the ${{\text{d}}_{xz}}$ lies in between the x and z-axis.
The orbital of ${{\text{d}}_{{{\text{z}}^{\text{2}}}}}$lies on the z-axis. So, the electron density of the ${{\text{d}}_{{{\text{z}}^{\text{2}}}}}$lies on the z-axis.
The orbitals of ${{\text{d}}_{{{\text{x}}^2} - {{\text{y}}^{\text{2}}}}}$ lie on the x and y-axis. So, the electron density of the ${{\text{d}}_{{{\text{x}}^2} - {{\text{y}}^{\text{2}}}}}$ lies on the x and y-axis.
So, ${{\text{d}}_{{{\text{z}}^{\text{2}}}}}{\text{,}}{{\text{d}}_{{{\text{x}}^2} - {{\text{y}}^{\text{2}}}}}$ pairs of d-orbitals will have electron density along the axis.
Therefore, option (A) ${{\text{d}}_{{{\text{z}}^{\text{2}}}}}{\text{,}}{{\text{d}}_{{{\text{x}}^2} - {{\text{y}}^{\text{2}}}}}$ is correct.
Note: Out of five d-orbitals, two lie on the axis and three lie in between the axis. So, when ligands form a complex with the metal it affects the three orbitals differently and two orbitals differently, so the five d-orbitals break into two parts of three and two.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Which among the following are examples of coming together class 11 social science CBSE




