Which of the following pairs are isostructural ?
(A) $SO_4^{2 - }$ and $BF_4^ - $
(B) $N{H_3}$ and $NH_4^ + $
(C) $CO_3^{2 - }$ and $C{O_2}$
(D) $C{H_4}$ and $B{F_3}$
Answer
581.4k+ views
Hint: Isostructural species are those which have the same shape and hybridization.
Number of election pair $ = \dfrac{1}{2}\left[ {N + V - C + A} \right]$
Where
$V = $ Number of valence electron present in central atom
$N = $ Number of non-monovalent atoms bonded to covalent atom
$C = $ Charge of cation
$A = $ Charge of anion
Hybridisation can be calculated by using this formula then we will be able to find the structure from hybridisation.
Complete step by step answer:
The given molecules are –
(A) $SO_4^{2 - }$ and $BF_4^ - $
Number of electron pair in $SO_4^{2 - } = \dfrac{1}{2} \times \left[ {6 + 0 + 2} \right] = 4$
Number of electron pairs are 4 that means the hybridisation will be $s{p^3}$ and the geometry of the molecule will be tetrahedral.
Number of electron pair in $BF_4^ - = \dfrac{1}{2} \times \left[ {3 + 4 + 1} \right] = 4$
Number of electron pair are 4 that means the hybridisation will be $s{p^3}$ and the geometry of molecule will be tetrahedral
Structures
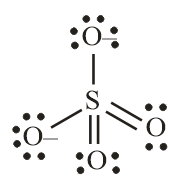
$SO_4^{2 - }$ molecule
(Tetrahedral structure)
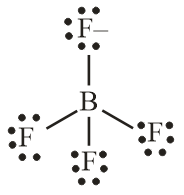
$BF_4^ - $ molecule
(Tetrahedral structure)
(B) $N{H_3}$ and $NH_4^ + $
Number of electron pair in $N{H_3} = \dfrac{1}{2} \times \left[ {5 + 3 + 0} \right] = 4$
Number of electron pairs are 4 that means the hybridisation will be $s{p^3}$ and geometry will be tetrahedral. But, In $N{H_3}$ Nitrogen is surrounded by 3 atoms and the fourth position will be occupied by lone pairs of electrons.
Hence, the structure of $N{H_3}$ will be pyramidal.
Number of electron pair in $NH_4^ + = \dfrac{1}{2}\left[ {5 + 4 - 1} \right] = 4$
Number of electron pairs are 4 that means the hybridisation will be $s{p^3}$ and the geometry will be tetrahedral.
Structures

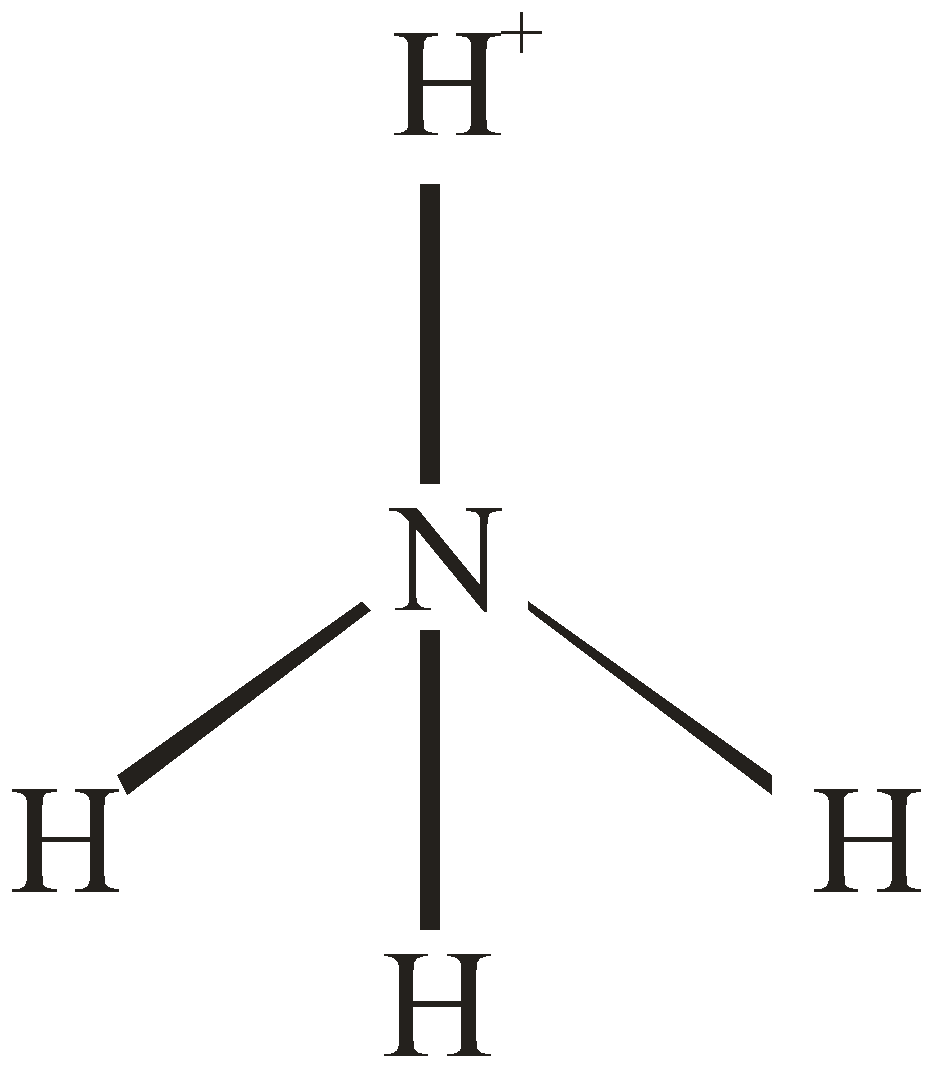
$N{H_3}$ molecule $NH_4^ + $ molecule
[Pyramidal structure] [Tetrahedral molecule]
(C) $CO_3^{2 - }$ and $C{O_2}$
Number of electron pair in $CO_3^{2 - } = \dfrac{1}{2}\left[ {4 + 0 + 2} \right] = 3$
The number of electron pairs are 3 that means the hybridisation will be $s{p^2}$ and the geometry will be trigonal planar.
Number of electron pair in $C{O_2} = \dfrac{1}{2}\left[ {4 + 0 + 0} \right] = 2$
Number of electron pairs are 2 that means the hybridisation will be sp and the geometry will be linear.
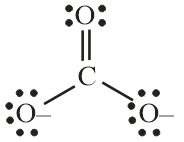
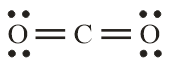
$CO_3^{2 - }$ molecule $C{O_2}$ molecule
(Trigonal planar structure) (Linear structure)
(D) $C{H_4}$ and $B{F_3}$
Number of electron pair in $C{H_4} = \dfrac{1}{2}\left[ {4 + 4 + 0} \right] = 4$
Number of electron pair are 4 that means the hybridisation will be $s{p^3}$ and the geometry will be tetrahedral
Number of electron pair in $B{F_3} = \dfrac{1}{2}\left[ {3 + 3 + 0} \right] = 3$
Number of electron pairs are 3 that means the hybridisation will be $s{p^3}$ and geometry will be trigonal planar.
Structures
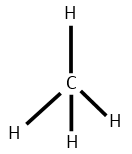
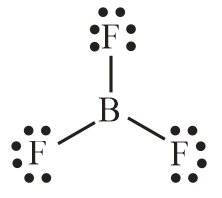
$C{H_4}$ molecule $B{F_3}$ molecule
(Tetrahedral structure) (Trigonal planar)
From this, we conclude that $SO_4^{2 - }$ and $BF_4^ - $ have the same structure.
Hence, the correct answer is (A) $SO_4^{2 - }$ and $BF_4^ - $
Note: Geometry of a molecule is the arrangement of lone pair and bond pair while shape is the molecular structure excluding lone pairs of central atoms.
Number of election pair $ = \dfrac{1}{2}\left[ {N + V - C + A} \right]$
Where
$V = $ Number of valence electron present in central atom
$N = $ Number of non-monovalent atoms bonded to covalent atom
$C = $ Charge of cation
$A = $ Charge of anion
Hybridisation can be calculated by using this formula then we will be able to find the structure from hybridisation.
Complete step by step answer:
The given molecules are –
(A) $SO_4^{2 - }$ and $BF_4^ - $
Number of electron pair in $SO_4^{2 - } = \dfrac{1}{2} \times \left[ {6 + 0 + 2} \right] = 4$
Number of electron pairs are 4 that means the hybridisation will be $s{p^3}$ and the geometry of the molecule will be tetrahedral.
Number of electron pair in $BF_4^ - = \dfrac{1}{2} \times \left[ {3 + 4 + 1} \right] = 4$
Number of electron pair are 4 that means the hybridisation will be $s{p^3}$ and the geometry of molecule will be tetrahedral
Structures

$SO_4^{2 - }$ molecule
(Tetrahedral structure)

$BF_4^ - $ molecule
(Tetrahedral structure)
(B) $N{H_3}$ and $NH_4^ + $
Number of electron pair in $N{H_3} = \dfrac{1}{2} \times \left[ {5 + 3 + 0} \right] = 4$
Number of electron pairs are 4 that means the hybridisation will be $s{p^3}$ and geometry will be tetrahedral. But, In $N{H_3}$ Nitrogen is surrounded by 3 atoms and the fourth position will be occupied by lone pairs of electrons.
Hence, the structure of $N{H_3}$ will be pyramidal.
Number of electron pair in $NH_4^ + = \dfrac{1}{2}\left[ {5 + 4 - 1} \right] = 4$
Number of electron pairs are 4 that means the hybridisation will be $s{p^3}$ and the geometry will be tetrahedral.
Structures


$N{H_3}$ molecule $NH_4^ + $ molecule
[Pyramidal structure] [Tetrahedral molecule]
(C) $CO_3^{2 - }$ and $C{O_2}$
Number of electron pair in $CO_3^{2 - } = \dfrac{1}{2}\left[ {4 + 0 + 2} \right] = 3$
The number of electron pairs are 3 that means the hybridisation will be $s{p^2}$ and the geometry will be trigonal planar.
Number of electron pair in $C{O_2} = \dfrac{1}{2}\left[ {4 + 0 + 0} \right] = 2$
Number of electron pairs are 2 that means the hybridisation will be sp and the geometry will be linear.

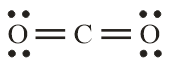
$CO_3^{2 - }$ molecule $C{O_2}$ molecule
(Trigonal planar structure) (Linear structure)
(D) $C{H_4}$ and $B{F_3}$
Number of electron pair in $C{H_4} = \dfrac{1}{2}\left[ {4 + 4 + 0} \right] = 4$
Number of electron pair are 4 that means the hybridisation will be $s{p^3}$ and the geometry will be tetrahedral
Number of electron pair in $B{F_3} = \dfrac{1}{2}\left[ {3 + 3 + 0} \right] = 3$
Number of electron pairs are 3 that means the hybridisation will be $s{p^3}$ and geometry will be trigonal planar.
Structures


$C{H_4}$ molecule $B{F_3}$ molecule
(Tetrahedral structure) (Trigonal planar)
From this, we conclude that $SO_4^{2 - }$ and $BF_4^ - $ have the same structure.
Hence, the correct answer is (A) $SO_4^{2 - }$ and $BF_4^ - $
Note: Geometry of a molecule is the arrangement of lone pair and bond pair while shape is the molecular structure excluding lone pairs of central atoms.
| S.No. | Molecules | Hybridization | Geometry | Molecular Structure |
| 1 | $C{H_4}$ | $s{p^3}$ | Tetrahedral | Tetrahedral |
| 2 | $N{H_3}$ | $s{p^3}$ | Tetrahedral | Pyramidal |
| 3 | ${H_2}O$ | $s{p^3}$ | Tetrahedral | V-shape or Angular |
| 4 | $B{F_3}$ | $s{p^2}$ | Trigonal planar | Trigonal planar |
| 5 | $Be{H_2}$ | sp | Linear | Linear |
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Which among the following are examples of coming together class 11 social science CBSE




