Which of the following overlaps is not present in $Xe{{O}_{3}}$ molecule?
A. $s{{p}^{3}}+{{p}_{x}}$
B. $s{{p}^{3}}+{{p}_{y}}$
C. ${{d}_{xz}}+{{p}_{x}}$
D. $s{{p}^{3}}+s$
Answer
608.7k+ views
Hint: Xenon forms two types of oxides. They are xenon trioxide and xenon tetroxide. The hybridization of xenon trioxide ($Xe{{O}_{3}}$ ) is $s{{p}^{3}}$ ). We have to draw an orbital diagram to know the overlapping of orbitals in the formation of xenon trioxide.
Complete answer:
- In the question it is asked to find which overlapping among the given options is not present in the xenon trioxide.
- Xenon belongs to group VIII A in the periodic table.
- Oxygen belongs to group VI A in the periodic table.
- The orbital overlapping and hybridization of the oxygen atoms with the xenon atom is as follows.
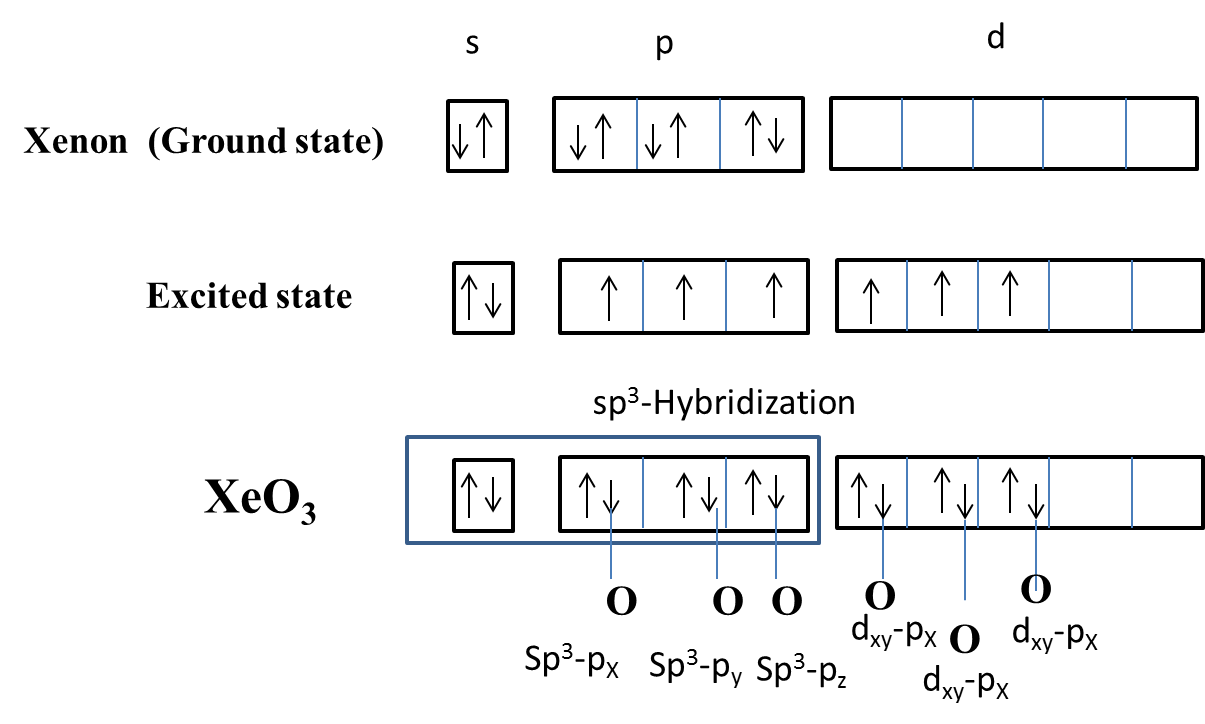
- From the above picture we can say that $s{{p}^{3}}+{{p}_{x}}$ , $s{{p}^{3}}+{{p}_{y}}$ and $s{{p}^{3}}+{{p}_{z}}$ are involved in the hybridization of xenon trioxide.
- Coming to option C, ${{d}_{xz}}+{{p}_{x}}$ is also involved in the hybridization process of xenon trioxide.
- We cannot see any $s{{p}^{3}}+s$ type of overlapping in the hybridization of the xenon trioxide.
- Therefore $s{{p}^{3}}+s$ type of overlapping is not present in xenon trioxide.
So, the correct option is D.
Note:
Xenon belongs to inert gases. Generally inert gases won’t react with other molecules. But in extreme conditions they react and form few derivatives as the products. Oxides of xenon belong to that category. The structure of xenon trioxide is trigonal pyramidal. In the structure of xenon trioxide there are three sigma bonds and three pi bonds. The oxidation state of xenon in xenon trioxide is ‘+6’.
Complete answer:
- In the question it is asked to find which overlapping among the given options is not present in the xenon trioxide.
- Xenon belongs to group VIII A in the periodic table.
- Oxygen belongs to group VI A in the periodic table.
- The orbital overlapping and hybridization of the oxygen atoms with the xenon atom is as follows.

- From the above picture we can say that $s{{p}^{3}}+{{p}_{x}}$ , $s{{p}^{3}}+{{p}_{y}}$ and $s{{p}^{3}}+{{p}_{z}}$ are involved in the hybridization of xenon trioxide.
- Coming to option C, ${{d}_{xz}}+{{p}_{x}}$ is also involved in the hybridization process of xenon trioxide.
- We cannot see any $s{{p}^{3}}+s$ type of overlapping in the hybridization of the xenon trioxide.
- Therefore $s{{p}^{3}}+s$ type of overlapping is not present in xenon trioxide.
So, the correct option is D.
Note:
Xenon belongs to inert gases. Generally inert gases won’t react with other molecules. But in extreme conditions they react and form few derivatives as the products. Oxides of xenon belong to that category. The structure of xenon trioxide is trigonal pyramidal. In the structure of xenon trioxide there are three sigma bonds and three pi bonds. The oxidation state of xenon in xenon trioxide is ‘+6’.
Recently Updated Pages
Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Master Class 9 Social Science: Engaging Questions & Answers for Success

Class 9 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




