Which of the following molecules has a tetrahedral shape?
(A)- ${\text{HgC}}{{\text{l}}_{\text{2}}}$
(B)- ${\text{C}}{{\text{O}}_{\text{2}}}$
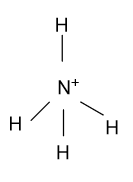
(C)- ${\text{NH}}_{\text{4}}^{\text{ + }}$
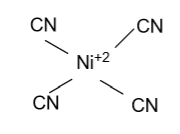
(D)- ${\text{Ni}}\left( {{\text{CN}}} \right)_{\text{4}}^{{\text{2 - }}}$
Answer
594.3k+ views
Hint: Molecule whose shape is tetrahedral, is having four hybrid orbitals and the hybridization of central atom in that molecule is ${\text{s}}{{\text{p}}^3}$ and bond angle between two bonds is around ${109^0}$.
Complete answer: In the above question following molecules are given and their hybridization is as follow:
-In ${\text{HgC}}{{\text{l}}_{\text{2}}}$, mercury is the central atom whose atomic number is 80 & two electrons are present in the outermost shell which binds with the 2 chlorine atoms and form ${\text{HgC}}{{\text{l}}_{\text{2}}}$ whose hybridization is ${\text{sp}}$ & having linear shape.
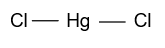
-In ${\text{C}}{{\text{O}}_{\text{2}}}$, carbon is the central atom whose atomic number is 6 & four electrons are present in the outermost shell which binds with the 2 oxygen atoms through double bond and form ${\text{C}}{{\text{O}}_{\text{2}}}$ whose hybridization is ${\text{sp}}$ & having linear shape.
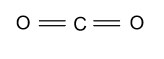
-In ${\text{NH}}_{\text{4}}^{\text{ + }}$, Nitrogen is the central atom whose atomic number is 7 & five electrons are present in the outermost shell but here nitrogen is present in +1 oxidation state it means nitrogen remove one electron from the outermost shell. Now the four electrons binds with the 4 hydrogen atoms and form ${\text{NH}}_{\text{4}}^{\text{ + }}$whose hybridization is ${\text{s}}{{\text{p}}^3}$ & having tetrahedral shape.
-In ${\text{Ni}}\left( {{\text{CN}}} \right)_{\text{4}}^{{\text{2 - }}}$, Nickel is the central atom whose atomic number is 28 & two electrons are present in the outermost shell but here nickel is present in +2 oxidation state it means Nickel removes two electrons from the outermost shell. In the given molecule ${\text{CN}}$ is the strong ligand who tries to pair all unpaired electrons of the d-orbital & we get one empty d-orbital. Now the four empty orbitals binds with the 4 ${\text{CN}}$ atoms and form ${\text{Ni}}\left( {{\text{CN}}} \right)_{\text{4}}^{{\text{2 - }}}$ whose hybridization is ${\text{ds}}{{\text{p}}^2}$ & having square planar shape.
Hence, option (C) is correct.
Note: In this question some of you may give the wrong answer by considering the option (D) molecule by keeping the fact that it is also making four bonds so that it will also show tetrahedral structure. But, that is not true because in the transition metal molecules we have to give attention to the ligand present on that molecule.
Complete answer: In the above question following molecules are given and their hybridization is as follow:
-In ${\text{HgC}}{{\text{l}}_{\text{2}}}$, mercury is the central atom whose atomic number is 80 & two electrons are present in the outermost shell which binds with the 2 chlorine atoms and form ${\text{HgC}}{{\text{l}}_{\text{2}}}$ whose hybridization is ${\text{sp}}$ & having linear shape.
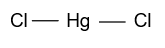
-In ${\text{C}}{{\text{O}}_{\text{2}}}$, carbon is the central atom whose atomic number is 6 & four electrons are present in the outermost shell which binds with the 2 oxygen atoms through double bond and form ${\text{C}}{{\text{O}}_{\text{2}}}$ whose hybridization is ${\text{sp}}$ & having linear shape.
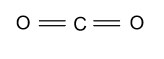
-In ${\text{NH}}_{\text{4}}^{\text{ + }}$, Nitrogen is the central atom whose atomic number is 7 & five electrons are present in the outermost shell but here nitrogen is present in +1 oxidation state it means nitrogen remove one electron from the outermost shell. Now the four electrons binds with the 4 hydrogen atoms and form ${\text{NH}}_{\text{4}}^{\text{ + }}$whose hybridization is ${\text{s}}{{\text{p}}^3}$ & having tetrahedral shape.

-In ${\text{Ni}}\left( {{\text{CN}}} \right)_{\text{4}}^{{\text{2 - }}}$, Nickel is the central atom whose atomic number is 28 & two electrons are present in the outermost shell but here nickel is present in +2 oxidation state it means Nickel removes two electrons from the outermost shell. In the given molecule ${\text{CN}}$ is the strong ligand who tries to pair all unpaired electrons of the d-orbital & we get one empty d-orbital. Now the four empty orbitals binds with the 4 ${\text{CN}}$ atoms and form ${\text{Ni}}\left( {{\text{CN}}} \right)_{\text{4}}^{{\text{2 - }}}$ whose hybridization is ${\text{ds}}{{\text{p}}^2}$ & having square planar shape.

Hence, option (C) is correct.
Note: In this question some of you may give the wrong answer by considering the option (D) molecule by keeping the fact that it is also making four bonds so that it will also show tetrahedral structure. But, that is not true because in the transition metal molecules we have to give attention to the ligand present on that molecule.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




