Which of the following molecules has a tetrahedral shape?
A. $HgC{l_2}$
B. $C{O_2}$
C. $N{H_4}^ + $
D. $Ni\left( {CN} \right)_4^{2 - }$
Answer
626.7k+ views
Hint: VSEPR helps in predicting the geometry of a compound taking in account the arrangement of electron pairs. VSEPR theory states that the electrons present around repel each other and it tends to take up an arrangement that will have minimum repulsion.The number of the valence shell present in the central metal atom is determined by drawing a Lewis structure of that atom.
Complete step by step answer:
$N{H_4}^ + $ has ${\rm{s}}{{\rm{p}}^{\rm{3}}}$ hybridization, that is, it involves one s atomic orbital and three p atomic orbital. It has a bond angle of ${109.5^0}$. N has the electronic configuration $\left[ {{\rm{He}}} \right]{\rm{2}}{{\rm{s}}^{\rm{2}}}{\rm{2}}{{\rm{p}}^{\rm{3}}}$ and hydrogen has the electronic configuration ${\rm{1}}{{\rm{s}}^{\rm{2}}}$. So, using this, we can now draw the tetrahedral structure of $N{H_4}^ + $ in the following way.
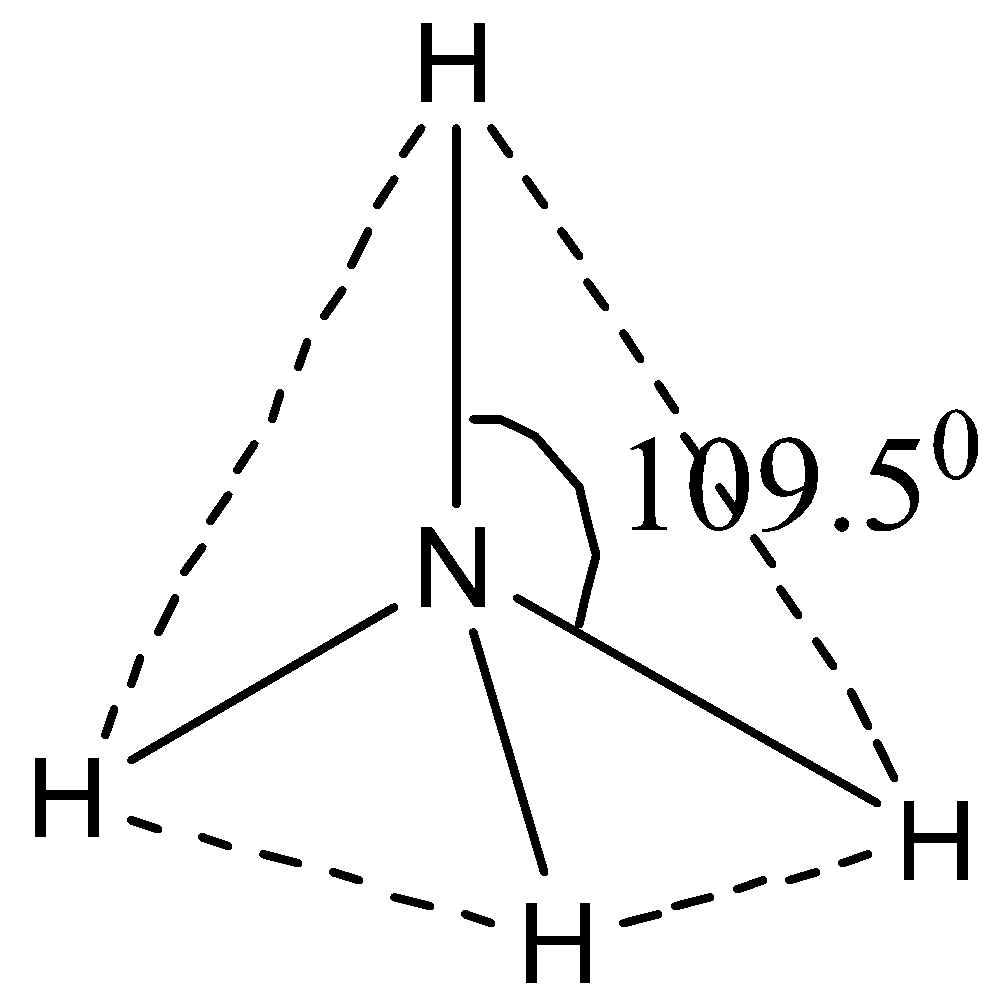
Therefore, $N{H_4}^ + $ it has a tetrahedral shape.
$HgC{l_2}$ and $C{O_2}$ involves sp hybridisation. Therefore, both of them have a linear structure.
$Ni\left( {CN} \right)_4^{2 - }$ has ${\rm{ds}}{{\rm{p}}^{\rm{2}}}$ hybridisation. It has a square planar geometry.
Hence C is the correct option.
Note:
The steric number of the central atom in the molecule is given as the number of atoms bonded to the central atom, which is called as the summation of the coordination number and the number of lone pairs of valence electrons on the central atom.
Complete step by step answer:
$N{H_4}^ + $ has ${\rm{s}}{{\rm{p}}^{\rm{3}}}$ hybridization, that is, it involves one s atomic orbital and three p atomic orbital. It has a bond angle of ${109.5^0}$. N has the electronic configuration $\left[ {{\rm{He}}} \right]{\rm{2}}{{\rm{s}}^{\rm{2}}}{\rm{2}}{{\rm{p}}^{\rm{3}}}$ and hydrogen has the electronic configuration ${\rm{1}}{{\rm{s}}^{\rm{2}}}$. So, using this, we can now draw the tetrahedral structure of $N{H_4}^ + $ in the following way.

Therefore, $N{H_4}^ + $ it has a tetrahedral shape.
$HgC{l_2}$ and $C{O_2}$ involves sp hybridisation. Therefore, both of them have a linear structure.
$Ni\left( {CN} \right)_4^{2 - }$ has ${\rm{ds}}{{\rm{p}}^{\rm{2}}}$ hybridisation. It has a square planar geometry.
Hence C is the correct option.
Note:
The steric number of the central atom in the molecule is given as the number of atoms bonded to the central atom, which is called as the summation of the coordination number and the number of lone pairs of valence electrons on the central atom.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

What are hard magnetic materials class 11 physics CBSE




