Which of the following molecule is linear:
(A) $S{{O}_{2}}$
(B) $N{{O}_{2}}^{+}$
(C) $N{{O}_{2}}$
(D) $SC{{l}_{2}}$
Answer
618k+ views
Hint: The 3-D arrangement of atoms in a molecule is known as its molecular geometry. The molecular geometry includes the shape of the molecule as well as the bond length and bond angles of the molecule. All these factors help to determine the position of each atom in the geometry.
Complete step by step solution:
VSEPR theory helps us to predict the geometry of the molecule from the number of electron pairs and lone pairs around the central atom.
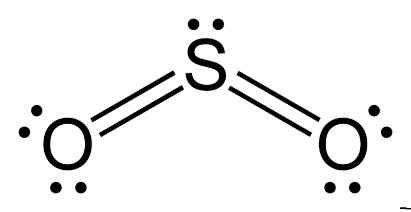
For $S{{O}_{2}}$
S has 6 unpaired electrons and oxygen is a divalent atom.
Hybridization can be calculated by adding sigma bond and lone pairs.
S has 1 lone pair and form 2 sigma bond: hybridization = $1+2=3$
And hybridization will be $s{{p}^{2}}$ and shape is v shaped
For $N{{O}_{2}}^{+}$
N has 5 unpaired electrons, due to positive charge N will have 4 unpaired electrons and oxygen is a divalent atom.
Hybridization can be calculated by adding sigma bonds and lone pairs.
N has 0 lone pair and form 2 sigma bond: hybridization = $0+2=2$
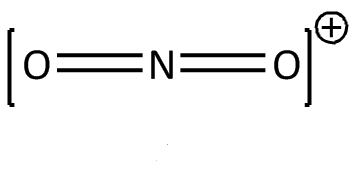
Hence the correct option is (B)
Note: Similarly for $N{{O}_{2}}$ hybridization will be $s{{p}^{2}}$ and shape is bent and for $SC{{l}_{2}}$ hybridization will be $s{{p}^{3}}$ and shape is bent .The geometry and shape of a molecule can be same or different as geometry of the molecule depends on the arrangement of lone pair and bond Pair while the shape of a molecule exclude the lone pair on the central atom.
Complete step by step solution:
VSEPR theory helps us to predict the geometry of the molecule from the number of electron pairs and lone pairs around the central atom.
For $S{{O}_{2}}$
S has 6 unpaired electrons and oxygen is a divalent atom.
Hybridization can be calculated by adding sigma bond and lone pairs.
S has 1 lone pair and form 2 sigma bond: hybridization = $1+2=3$
And hybridization will be $s{{p}^{2}}$ and shape is v shaped

For $N{{O}_{2}}^{+}$
N has 5 unpaired electrons, due to positive charge N will have 4 unpaired electrons and oxygen is a divalent atom.
Hybridization can be calculated by adding sigma bonds and lone pairs.
N has 0 lone pair and form 2 sigma bond: hybridization = $0+2=2$
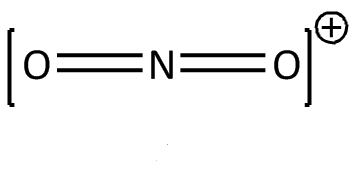
Hence the correct option is (B)
Note: Similarly for $N{{O}_{2}}$ hybridization will be $s{{p}^{2}}$ and shape is bent and for $SC{{l}_{2}}$ hybridization will be $s{{p}^{3}}$ and shape is bent .The geometry and shape of a molecule can be same or different as geometry of the molecule depends on the arrangement of lone pair and bond Pair while the shape of a molecule exclude the lone pair on the central atom.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Draw a diagram showing the external features of fish class 11 biology CBSE

What organs are located on the left side of your body class 11 biology CBSE

Difference between physical and chemical change class 11 chemistry CBSE

What is the weight in lbs of a 75 Kg person class 11 physics CBSE

Which of the following is the hottest month AMay BJune class 11 social science CBSE




