Which of the following is not a Lewis acid?
(a)- $FeC{{l}_{3}}$
(b)- $BC{{l}_{3}}$
(c)- $ZnC{{l}_{2}}$
(d)- $N{{H}_{3}}$
Answer
600.3k+ views
Hint: Lewis explained that an acid accepts a pair of electrons and the base donates a pair of electrons. So, we can identify the compound which has electrons to donate is not a Lewis acid.
Complete step by step answer:
According to Lewis, an acid can be defined as a substance that can accept a pair of the electron, and a base is that substance that can donate a pair of electrons.
So, the acid must be an electron-deficient compound and the base must be an electron-rich compound.
In $FeC{{l}_{3}}$, the iron atom has an incomplete octet. The iron atom is bonded with three chlorine atoms with electrons and it can accept a pair of the electron, therefore it is a Lewis acid.
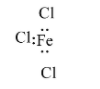
In $BC{{l}_{3}}$, the boron atom has an incomplete octet. The boron atom is bonded with three chlorine atoms with electrons and it can accept a pair of the electron, therefore it is a Lewis acid.

In $ZnC{{l}_{2}}$, the zinc atom has an incomplete octet. The zinc atom is bonded with two chlorine atoms, this means that zinc is in +2 oxidation state, hence it can accept a pair of electrons. Therefore, it is a Lewis acid.

In $N{{H}_{3}}$, the nitrogen atom is bonded with three hydrogen atoms, but the valence electrons in nitrogen are 5. So it has a pair of electrons that are non-bonded. So, it donates a pair of electrons. It is a Lewis base.
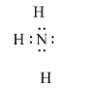
So, the correct answer is an option (d)- $N{{H}_{3}}$
Note: Some other examples of Lewis acid in molecular form are $B{{F}_{3}}$. Cations that acts as Lewis acid are $A{{g}^{+}},C{{u}^{2+}},F{{e}^{3+}}$, etc. The molecules having empty d-orbital also act as Lewis acid, $Si{{F}_{4}}$.
Complete step by step answer:
According to Lewis, an acid can be defined as a substance that can accept a pair of the electron, and a base is that substance that can donate a pair of electrons.
So, the acid must be an electron-deficient compound and the base must be an electron-rich compound.
In $FeC{{l}_{3}}$, the iron atom has an incomplete octet. The iron atom is bonded with three chlorine atoms with electrons and it can accept a pair of the electron, therefore it is a Lewis acid.
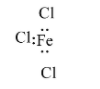
In $BC{{l}_{3}}$, the boron atom has an incomplete octet. The boron atom is bonded with three chlorine atoms with electrons and it can accept a pair of the electron, therefore it is a Lewis acid.

In $ZnC{{l}_{2}}$, the zinc atom has an incomplete octet. The zinc atom is bonded with two chlorine atoms, this means that zinc is in +2 oxidation state, hence it can accept a pair of electrons. Therefore, it is a Lewis acid.

In $N{{H}_{3}}$, the nitrogen atom is bonded with three hydrogen atoms, but the valence electrons in nitrogen are 5. So it has a pair of electrons that are non-bonded. So, it donates a pair of electrons. It is a Lewis base.
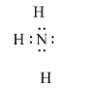
So, the correct answer is an option (d)- $N{{H}_{3}}$
Note: Some other examples of Lewis acid in molecular form are $B{{F}_{3}}$. Cations that acts as Lewis acid are $A{{g}^{+}},C{{u}^{2+}},F{{e}^{3+}}$, etc. The molecules having empty d-orbital also act as Lewis acid, $Si{{F}_{4}}$.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Discuss the various forms of bacteria class 11 biology CBSE




