Which of the following is anti aromatic in nature
A.
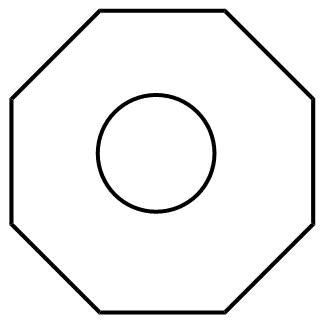
C.
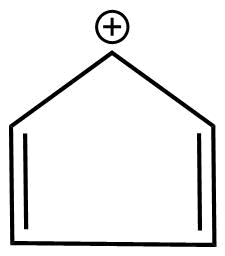
D.
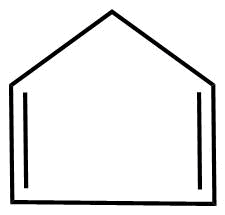
E.





Answer
588k+ views
Hint: A compound which has planar conjugated cyclic system and contains $4n$ pi-electrons which are destabilized by resonance are known as anti aromatic compounds .
Complete step by step answer:
We need four criterias to define Aromaticity and no exceptions are allowed.
1. the molecule has to be cyclic
2. Every atom in the ring has to be in conjugation, or every atom should have a p orbital.
3. the molecule should follow huckle’s rule for aromaticity
\[4n + 2\]
Where, $n$ is an integer
If it does not follow huckel's rule, check for $4n$ rule, if it satisfies this rule, then the molecule is non aromatic.
Where, $n$ is an integer
4. The molecule should be planar in nature.
Now, let us review the options one by one by checking off every point.
A.
1. The molecules are cyclic.
2. Every atom in the ring is in conjugation
3.It does not obey huckel's rule
But it does obey $4n$ rule
Where, $n = 2$
3. It is planar in nature.
Hence, the molecule is non-aromatic.
B.
1. The molecules are cyclic.
2. Every atom in the ring is not conjugation due to the positive charge there is discontinuation .
3.It does not obey huckel's rule
3. It is planar in nature.
Hence, the molecule is antiaromatic.
C. 1. The molecules are cyclic.
2. Every atom in the ring is in conjugation
3.It does not obey huckel's rule
But it does obey $4n$ rule
Where, $n = 2$
3. It is planar in nature.
Hence, the molecule is non-aromatic.
D.
1. The molecules are cyclic.
2. Every atom in the ring is in conjugation
3.It does obey huckel's rule
Where, $n = 2$
3. It is planar in nature.
Hence, the molecule is aromatic.
So, the correct answer is Option B.
Note: Compounds which do not obey Huckel’s rule are termed as anti aromatic compounds and are not stable compounds .They are even less stable than the corresponding non-planar systems.
Complete step by step answer:
We need four criterias to define Aromaticity and no exceptions are allowed.
1. the molecule has to be cyclic
2. Every atom in the ring has to be in conjugation, or every atom should have a p orbital.
3. the molecule should follow huckle’s rule for aromaticity
\[4n + 2\]
Where, $n$ is an integer
If it does not follow huckel's rule, check for $4n$ rule, if it satisfies this rule, then the molecule is non aromatic.
Where, $n$ is an integer
4. The molecule should be planar in nature.
Now, let us review the options one by one by checking off every point.
A.
1. The molecules are cyclic.
2. Every atom in the ring is in conjugation
3.It does not obey huckel's rule
But it does obey $4n$ rule
Where, $n = 2$
3. It is planar in nature.
Hence, the molecule is non-aromatic.
B.
1. The molecules are cyclic.
2. Every atom in the ring is not conjugation due to the positive charge there is discontinuation .
3.It does not obey huckel's rule
3. It is planar in nature.
Hence, the molecule is antiaromatic.
C. 1. The molecules are cyclic.
2. Every atom in the ring is in conjugation
3.It does not obey huckel's rule
But it does obey $4n$ rule
Where, $n = 2$
3. It is planar in nature.
Hence, the molecule is non-aromatic.
D.
1. The molecules are cyclic.
2. Every atom in the ring is in conjugation
3.It does obey huckel's rule
Where, $n = 2$
3. It is planar in nature.
Hence, the molecule is aromatic.
So, the correct answer is Option B.
Note: Compounds which do not obey Huckel’s rule are termed as anti aromatic compounds and are not stable compounds .They are even less stable than the corresponding non-planar systems.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Explain zero factorial class 11 maths CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




