Which of the following is an aromatic compound?
A. Phenol
B. Naphthalene
C. Pyridine
D. All of the above
Answer
574.8k+ views
Hint: Aromatic compounds are also known by another name aromatic or arenes. These are categorized into the hydrocarbons having sigma bonds and pi electrons delocalised between carbon atoms in a ring. The most important condition for aromatic compounds is to satisfy the Huckel rule. Benzene is an example of an aromatic compound.
Complete step by step answer:
Let us discuss the most important condition mentioned for aromatic compounds, i.e. Huckel rule.
According to the Huckel rule, a compound will be aromatic if it satisfies the below- mentioned characteristics:
1.The compound should be planar
2.There should be complete delocalization of pi (\[\pi \]) electrons in the ring
3.There must be the presence of $(4n + 2)\pi $ electrons in the ring, where n is an integer.
Now, we will look at the given options one by one.
Phenol
The structure of phenol is as follows:
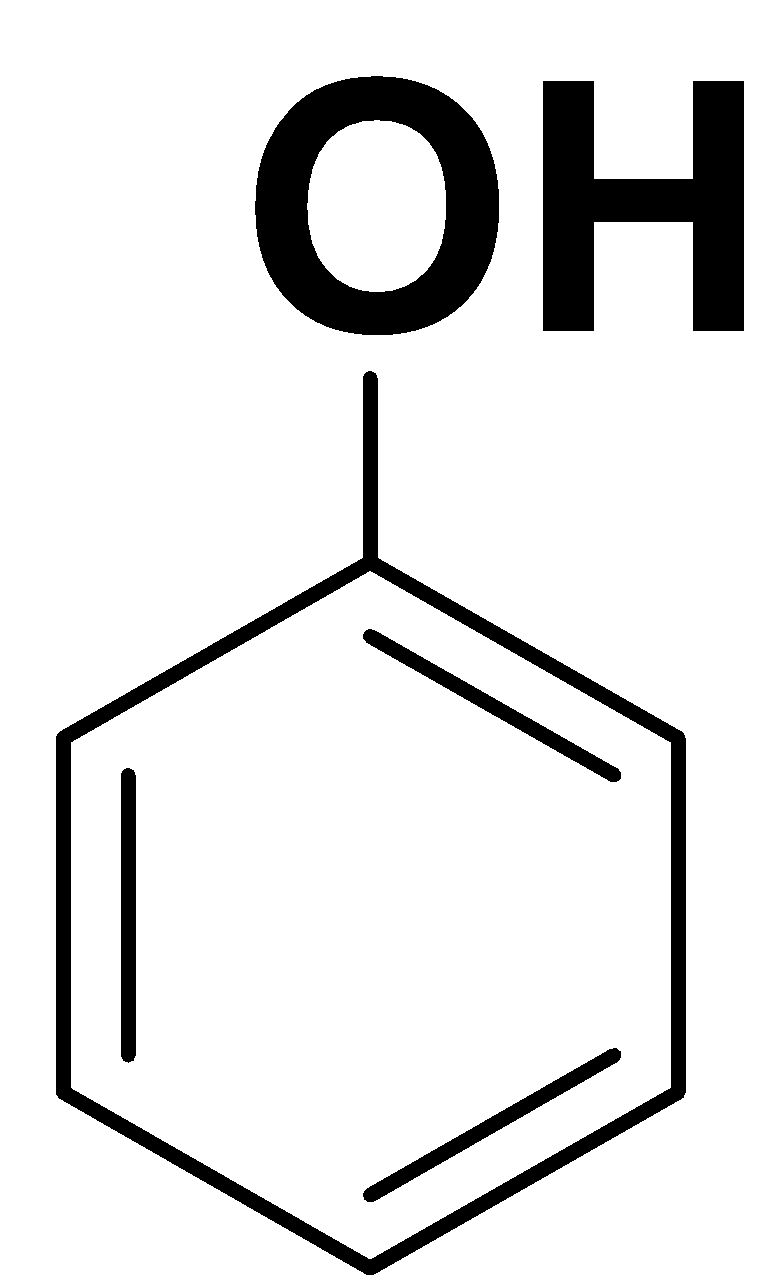
Here, we can see that the compound is planar and there is complete delocalization of pi electrons in the ring. Here, n is 1. So,
$n = 1$
$ \Rightarrow (4 \times 1 + 2)\pi $
$ \Rightarrow 6\pi $ electrons
In the phenol, alkoxide ion (${O^ - }$) is delocalized over the benzene ring. Thus, phenol is an aromatic compound.
Naphthalene
The structure of naphthalene is as follows:
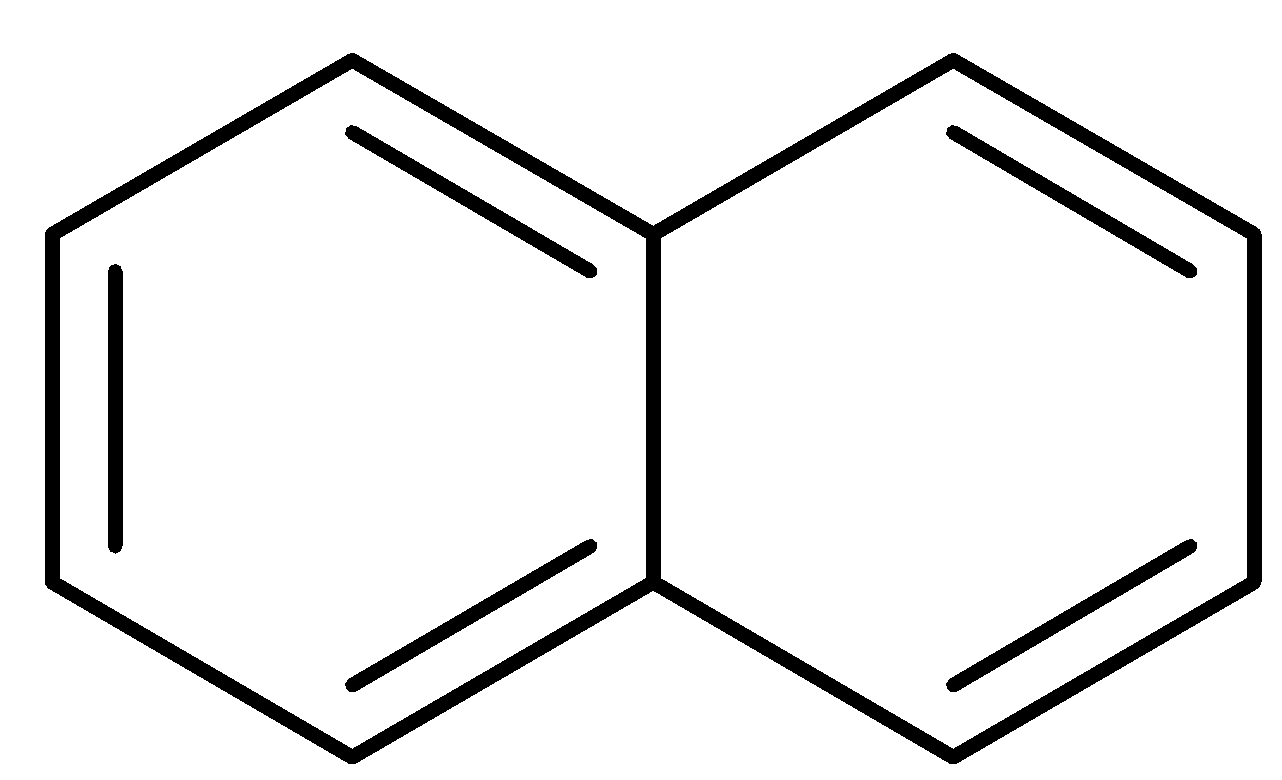
Similarly, like phenol, naphthalene is also planar and exhibits complete delocalization of pi electrons. Here, n is 2. So,
$n = 2$
$ \Rightarrow (4 \times 2 + 2)\pi $
$ \Rightarrow 10\pi $ electrons
Thus, naphthalene is an aromatic compound.
Pyridine
The structure of pyridine is as follows:
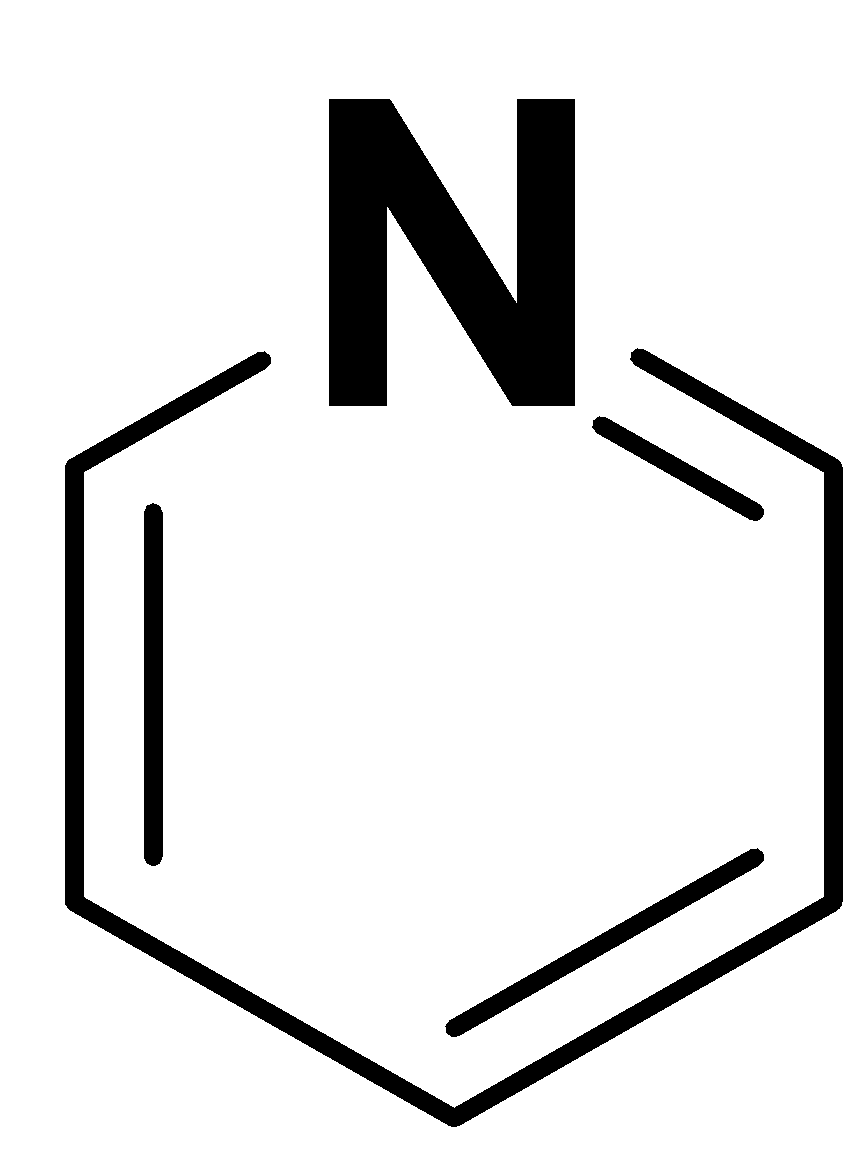
Pyridine is a six-membered ring with heteroatom i.e. nitrogen. It is not based on a benzene ring. So, we can say that it is an example of heteroarenes.
Pyridine follows the $(4n + 2)\pi $ condition.
So, it is an aromatic compound.
In the last, we can conclude that phenol, naphthalene and pyridine are aromatic compounds.
So, the correct answer is Option D.
Note: Aromatic compounds are categorized further into two categories i.e. Benzenoids and non-Benzenoids. Benzenoids are the compounds having at least one benzene ring. For example, Phenol etc. Non- Benzenoids are the compounds which don’t have benzene rings. In that one carbon is replaced with heteroatoms like sulphur, nitrogen etc. For example, pyridine. Non-Benzenoids are also known as heteroarenes.
Complete step by step answer:
Let us discuss the most important condition mentioned for aromatic compounds, i.e. Huckel rule.
According to the Huckel rule, a compound will be aromatic if it satisfies the below- mentioned characteristics:
1.The compound should be planar
2.There should be complete delocalization of pi (\[\pi \]) electrons in the ring
3.There must be the presence of $(4n + 2)\pi $ electrons in the ring, where n is an integer.
Now, we will look at the given options one by one.
Phenol
The structure of phenol is as follows:

Here, we can see that the compound is planar and there is complete delocalization of pi electrons in the ring. Here, n is 1. So,
$n = 1$
$ \Rightarrow (4 \times 1 + 2)\pi $
$ \Rightarrow 6\pi $ electrons
In the phenol, alkoxide ion (${O^ - }$) is delocalized over the benzene ring. Thus, phenol is an aromatic compound.
Naphthalene
The structure of naphthalene is as follows:

Similarly, like phenol, naphthalene is also planar and exhibits complete delocalization of pi electrons. Here, n is 2. So,
$n = 2$
$ \Rightarrow (4 \times 2 + 2)\pi $
$ \Rightarrow 10\pi $ electrons
Thus, naphthalene is an aromatic compound.
Pyridine
The structure of pyridine is as follows:

Pyridine is a six-membered ring with heteroatom i.e. nitrogen. It is not based on a benzene ring. So, we can say that it is an example of heteroarenes.
Pyridine follows the $(4n + 2)\pi $ condition.
So, it is an aromatic compound.
In the last, we can conclude that phenol, naphthalene and pyridine are aromatic compounds.
So, the correct answer is Option D.
Note: Aromatic compounds are categorized further into two categories i.e. Benzenoids and non-Benzenoids. Benzenoids are the compounds having at least one benzene ring. For example, Phenol etc. Non- Benzenoids are the compounds which don’t have benzene rings. In that one carbon is replaced with heteroatoms like sulphur, nitrogen etc. For example, pyridine. Non-Benzenoids are also known as heteroarenes.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Discuss the various forms of bacteria class 11 biology CBSE




