
Which of the following compounds might be useful to the chemist trying to increase the optical purity of (d) sample?
(a)
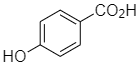
(b)
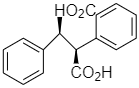
(c)
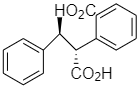
(d)
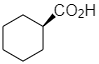




Answer
510.6k+ views
Hint: Substances which rotate the plane of polarized light are optically active substances. The isomer which rotates the plane of polarized light to the right is called dextro rotatory designated as ( $d$ ) and the one which rotates the plane of polarized light to the left is called laevo rotatory designated as $(l)$.
Complete answer:
Optical purity is a measure of its enantiomeric purity. Optical purity explains how much of one enantiomer is present in excess in the racemic mixture. Enantiomeric molecules rotate the plane in opposite directions but with the same magnitude. Optically active isomers are mirror image compounds and non-superimposable on each other and do not possess the plane of symmetry.
These optical isomers also have the property of Chirality. The necessary condition for a substance to show optical activity is that the substance should not have a plane of symmetry in its structure.
Optically active compounds are used for resolution of $ \pm $ mixture.
Option (b) is optically active
And the other options present are optically inactive.
meso compound compounds have chiral centres but they do not show optical activity due to internal compensation, It is superimposed on its mirror image.
Therefore the correct answer is option (b).
Note:
A $1:1$ equilibrium mixture of $d{\text{ }}$ and $l$ isomers gives a net zero rotation and is called a racemic mixture. A racemate is optically inactive as there is no net rotation of plane-polarised light.
However the enantiomers rotate plane-polarised light in opposite directions, the rotations cancel out because they are present in equal amounts.
Complete answer:
Optical purity is a measure of its enantiomeric purity. Optical purity explains how much of one enantiomer is present in excess in the racemic mixture. Enantiomeric molecules rotate the plane in opposite directions but with the same magnitude. Optically active isomers are mirror image compounds and non-superimposable on each other and do not possess the plane of symmetry.
These optical isomers also have the property of Chirality. The necessary condition for a substance to show optical activity is that the substance should not have a plane of symmetry in its structure.
Optically active compounds are used for resolution of $ \pm $ mixture.
Option (b) is optically active
And the other options present are optically inactive.
meso compound compounds have chiral centres but they do not show optical activity due to internal compensation, It is superimposed on its mirror image.
Therefore the correct answer is option (b).
Note:
A $1:1$ equilibrium mixture of $d{\text{ }}$ and $l$ isomers gives a net zero rotation and is called a racemic mixture. A racemate is optically inactive as there is no net rotation of plane-polarised light.
However the enantiomers rotate plane-polarised light in opposite directions, the rotations cancel out because they are present in equal amounts.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

Explain zero factorial class 11 maths CBSE




