
Which of the following compounds has a P−P bond?
(A) ${{\left( HP{{O}_{3}} \right)}_{3}}$
(B) ${{H}_{4}}{{P}_{2}}{{O}_{6}}$
(C) ${{H}_{4}}{{P}_{2}}{{O}_{7}}$
(D) ${{H}_{4}}{{P}_{2}}{{O}_{5}}$
Answer
589.8k+ views
Hint: In a P−P bond two phosphorous atoms are bonded with each other and form a bridge between the atoms. The bond between two phosphorus atoms is a covalent bond which is formed by the sharing of electrons by different atoms in a molecule.
Complete step by step solution:
- Among the given options ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ contains the P−P bond. The chemical name of${{H}_{4}}{{P}_{2}}{{O}_{6}}$ is hypophosphoric acid. The acid has an oxidation state of +4. The structure of hypophosphoric acid is given below
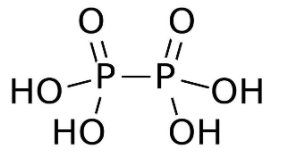
- As we can see in the structure of ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ a single P−P bond is present. Also it contains two P=O bonds and four P−OH bonds .In the structure of ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ the P−O bonds have a bond length of 151 pm and P−P bond has a bond length of 219 pm.
- ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ is a mineral acid and in the solid state it is present as dihydrate which has the molecular formula${{H}_{4}}{{P}_{2}}{{O}_{6}}.2{{H}_{2}}O$. It contains oxonium ions and is formulated as${{\left[ {{H}_{3}}{{O}^{+}} \right]}_{_{2}}}{{\left[ {{H}_{2}}{{P}_{2}}{{O}_{6}} \right]}^{2-}}$.
From the above discussions it’s clear that the compound hypophosphoric acid ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ has a P−P bond in it.
Therefore, the answer is option (B). ${{H}_{4}}{{P}_{2}}{{O}_{6}}$.
Additional information:
The hypophosphoric acid is generally used as reducing agents, wetting agents, as a bleaching agent and as a stimulant in pharmaceutical agents.
Note: Keep in mind that the hypophosphoric acid is a triprotic acid. Also do not confuse Hypophosphorous acid with Hypophosphoric acid. Hypophosphorous acid or phosphinic acid has the molecular formula ${{H}_{3}}P{{O}_{2}}$ whereas Hypophosphoric acid has the molecular formula ${{H}_{4}}{{P}_{2}}{{O}_{6}}$. In addition to this, there are also other oxyacids of phosphorus such as Phosphorus acid, Diphosphoric acid (Pyrophosphoric acid), Peroxophosphoric acid etc.
Complete step by step solution:
- Among the given options ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ contains the P−P bond. The chemical name of${{H}_{4}}{{P}_{2}}{{O}_{6}}$ is hypophosphoric acid. The acid has an oxidation state of +4. The structure of hypophosphoric acid is given below

- As we can see in the structure of ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ a single P−P bond is present. Also it contains two P=O bonds and four P−OH bonds .In the structure of ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ the P−O bonds have a bond length of 151 pm and P−P bond has a bond length of 219 pm.
- ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ is a mineral acid and in the solid state it is present as dihydrate which has the molecular formula${{H}_{4}}{{P}_{2}}{{O}_{6}}.2{{H}_{2}}O$. It contains oxonium ions and is formulated as${{\left[ {{H}_{3}}{{O}^{+}} \right]}_{_{2}}}{{\left[ {{H}_{2}}{{P}_{2}}{{O}_{6}} \right]}^{2-}}$.
From the above discussions it’s clear that the compound hypophosphoric acid ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ has a P−P bond in it.
Therefore, the answer is option (B). ${{H}_{4}}{{P}_{2}}{{O}_{6}}$.
Additional information:
The hypophosphoric acid is generally used as reducing agents, wetting agents, as a bleaching agent and as a stimulant in pharmaceutical agents.
Note: Keep in mind that the hypophosphoric acid is a triprotic acid. Also do not confuse Hypophosphorous acid with Hypophosphoric acid. Hypophosphorous acid or phosphinic acid has the molecular formula ${{H}_{3}}P{{O}_{2}}$ whereas Hypophosphoric acid has the molecular formula ${{H}_{4}}{{P}_{2}}{{O}_{6}}$. In addition to this, there are also other oxyacids of phosphorus such as Phosphorus acid, Diphosphoric acid (Pyrophosphoric acid), Peroxophosphoric acid etc.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

The largest wind power cluster is located in the state class 11 social science CBSE

Explain zero factorial class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Which among the following are examples of coming together class 11 social science CBSE

Can anyone list 10 advantages and disadvantages of friction




