Which of the following annulenes are aromatic?
A.[8]-Annulene
B.[10]-Annulene
C.[12]-Annulene
D.None
Answer
557k+ views
Hint: If a molecule is aromatic, then it should fulfil these conditions: 1. It should be cyclic, planar and has continuous delocalization of \[\pi \] electrons (electrons in p orbitals) and the delocalized π-electron cloud must contain a total of \[\left( {4n + 2} \right)\pi \] electrons (Huckel’s rule), where n is a whole number (i.e., n =0, 1, 2, 3 and so on).
Complete step by step answer:
Annulenes are the family of completely conjugated, monocyclic hydrocarbons containing alternating ring double bonds, such as benzene, but of different sizes. They have a general formula of ${C_n}{H_n}$ , where ‘n’ is an even number or ${C_n}{H_{n + 1}}$ , where n is an odd number.
The annulenes are named as [n]-annulene where n is an even number and it represents the number of Carbon atoms in the ring.
For example: Benzene is [6]-Annulene, cyclobutadiene is [4]-Annulene, cyclooctatetraene is [8]-Annulene.
Annulenes can be aromatic, anti-aromatic or non-aromatic. The behaviour of the annulenes can be explained on the basis of Huckel’s rule ( \[\left( {4n + 2} \right)\pi \] electrons).
[8]-Annulene is also known as 1,3,5,7-cyclooctatetraene. It has a planar cyclic conjugated system which has 4nπ electrons, where \[n = 2\] . To overcome the strain molecule assumes a non-planar, tub-shaped geometry. So, it is non-aromatic
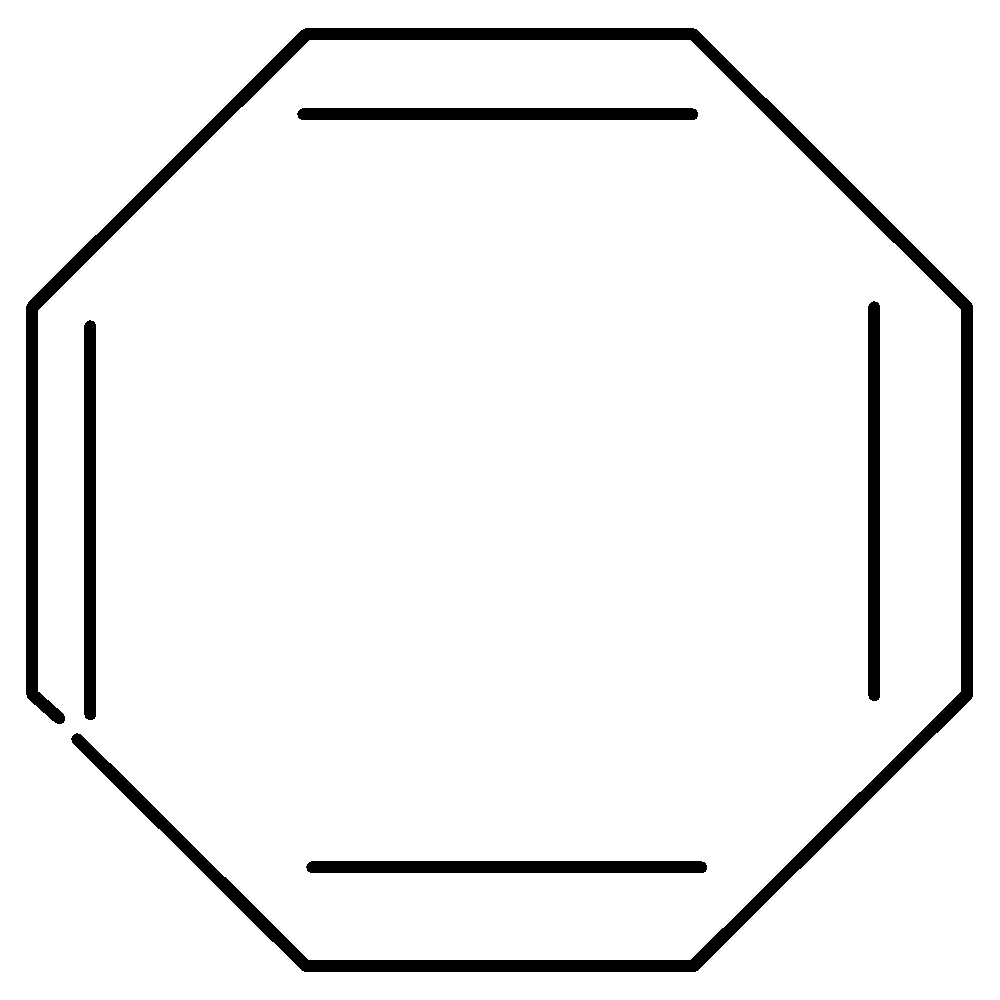
Figure: [8]-Annulene
[10]-Annulene is not particularly stable due to geometric factors. It possesses \[10\pi \] electrons so it gives an indication of aromaticity as per the \[\left( {4n + 2} \right)\pi \] electrons requirement for Huckel’s rule but it is not aromatic because various types of ring strain destabilize an all-planar geometry. So, it is unable to adopt the necessary planar configuration.
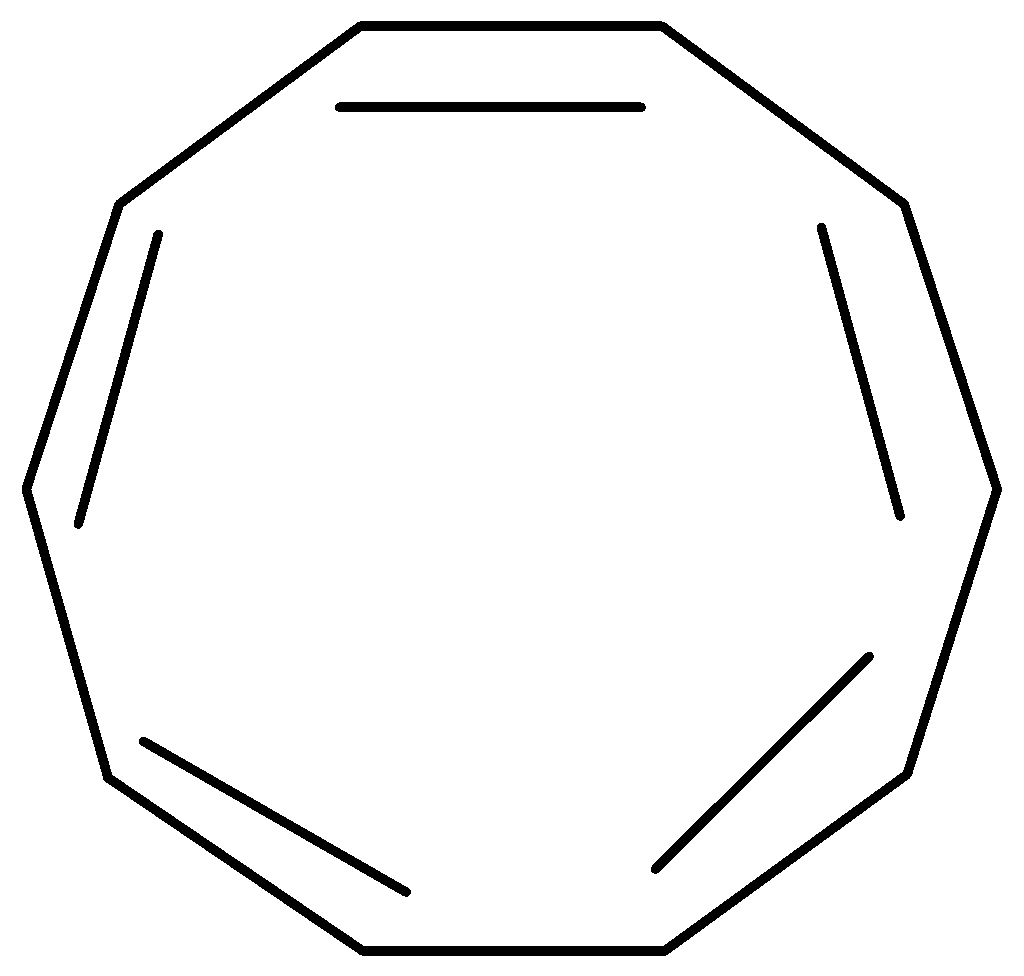
Figure: [10]-Annulene
In [12]-Annulene, the three H in-between the ring is far enough and do not create any strain for the planar arrangement. So, this is a cyclic, planar system having continuous delocalisation of π electrons and fulfilling the first condition. But the number of π electrons continuously delocalised are 12 i.e. \[4n\pi \] electrons, where \[n = 3\] . Since it is a \[4n\pi \] electron system, it is anti-aromatic in nature.
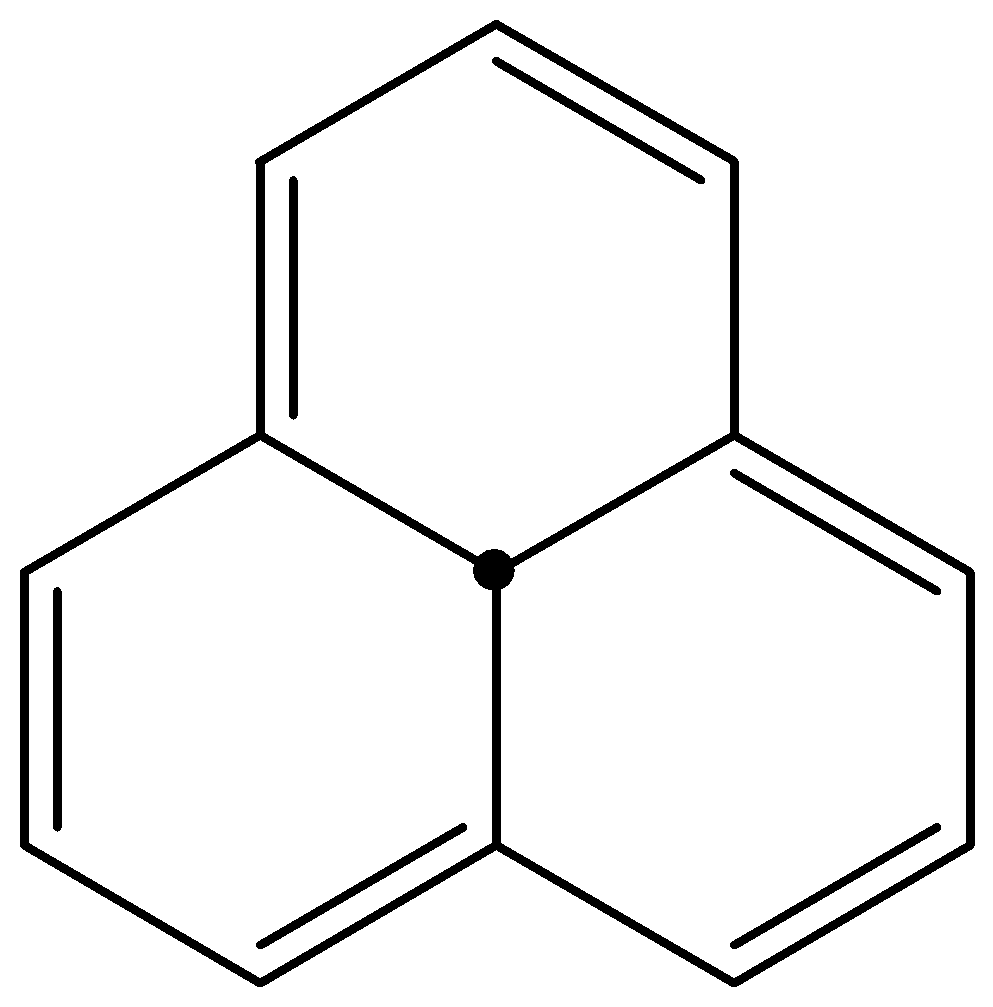
Figure: [12]-Annulene
Therefore, the correct answer is option (D).
Note: In [8]-Annulene, the regular planar octagon has bond angles of \[135^\circ \] with large bond angle strain due to large deviation from $s{p_2}$ bond angles of \[120^\circ \] and to overcome the strain, molecule assumes a non-planar, tub-shaped geometry. Hence, it is non-aromatic.
Complete step by step answer:
Annulenes are the family of completely conjugated, monocyclic hydrocarbons containing alternating ring double bonds, such as benzene, but of different sizes. They have a general formula of ${C_n}{H_n}$ , where ‘n’ is an even number or ${C_n}{H_{n + 1}}$ , where n is an odd number.
The annulenes are named as [n]-annulene where n is an even number and it represents the number of Carbon atoms in the ring.
For example: Benzene is [6]-Annulene, cyclobutadiene is [4]-Annulene, cyclooctatetraene is [8]-Annulene.
Annulenes can be aromatic, anti-aromatic or non-aromatic. The behaviour of the annulenes can be explained on the basis of Huckel’s rule ( \[\left( {4n + 2} \right)\pi \] electrons).
[8]-Annulene is also known as 1,3,5,7-cyclooctatetraene. It has a planar cyclic conjugated system which has 4nπ electrons, where \[n = 2\] . To overcome the strain molecule assumes a non-planar, tub-shaped geometry. So, it is non-aromatic

Figure: [8]-Annulene
[10]-Annulene is not particularly stable due to geometric factors. It possesses \[10\pi \] electrons so it gives an indication of aromaticity as per the \[\left( {4n + 2} \right)\pi \] electrons requirement for Huckel’s rule but it is not aromatic because various types of ring strain destabilize an all-planar geometry. So, it is unable to adopt the necessary planar configuration.

Figure: [10]-Annulene
In [12]-Annulene, the three H in-between the ring is far enough and do not create any strain for the planar arrangement. So, this is a cyclic, planar system having continuous delocalisation of π electrons and fulfilling the first condition. But the number of π electrons continuously delocalised are 12 i.e. \[4n\pi \] electrons, where \[n = 3\] . Since it is a \[4n\pi \] electron system, it is anti-aromatic in nature.

Figure: [12]-Annulene
Therefore, the correct answer is option (D).
Note: In [8]-Annulene, the regular planar octagon has bond angles of \[135^\circ \] with large bond angle strain due to large deviation from $s{p_2}$ bond angles of \[120^\circ \] and to overcome the strain, molecule assumes a non-planar, tub-shaped geometry. Hence, it is non-aromatic.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Explain zero factorial class 11 maths CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




