Which is most acidic and why?
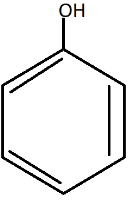
A.
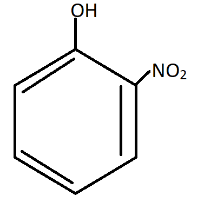
B.
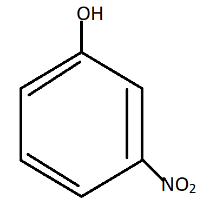
C.
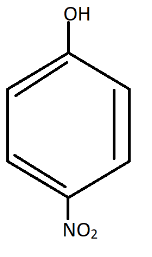
D.




Answer
524.4k+ views
Hint: Use the concept of inductive effect and mesomeric effect. Compare the inductive effect of the $N{O_2}$ group at different positions. Hydrogen bonding also plays an important role while comparing the acidity of compounds. It can be intramolecular hydrogen bonding.
Complete answer: Let us suppose that ${H^ + }$from each compound is removed. Then, the remaining $Oxide{\text{ ion}}$ \[\left( {{O^ - }} \right)\]must be stabilized by the Nitro group.
$N{O_2}$ group has both $\left( { - I} \right)$and $\left( { - m} \right)$effect. Hence, it supports the acidic nature of compounds. With the help of $\left( { - m} \right)$effect, the conjugate base $\left( {Oxide{\text{ ion}}} \right)$gets stabilized after removal of ${H^ + }$.
Nitro is an electron withdrawing group. It will withdraw electrons of $Oxide{\text{ ion}}$\[\left( {{O^ - }} \right)\]through these two effects.
By inductive effect, it withdraws electrons, but it is an effect which is based on distance unlike mesomeric effect.
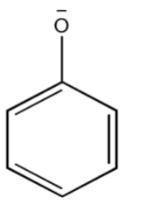
Thus, the $phenoxide{\text{ ion}}$so formed after losing the proton ${H^ + }$must be stabilized by resonance. The negative charge present on $phenoxide{\text{ ion}}$must be delocalized into a benzene ring.
Nitro group present at meta-position in$m - nitrophenol$, cannot stabilize $phenoxide{\text{ ion}}$as compared to $o - nitrophenol$ and$p - nitrophenol$.
Also, in$p - nitrophenol$, there is a chance of hydrogen bonding. (Hydrogen from $OH$ and Nitrogen from $N{O_2}$ group).
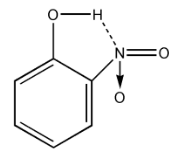
So, it is hard to lose the proton ${H^ + }$ due to $H - Bonding$ with $N{O_2}$ group.
Now, there will be more resonance effect, thus more stabilization of compound$p - nitrophenol$.
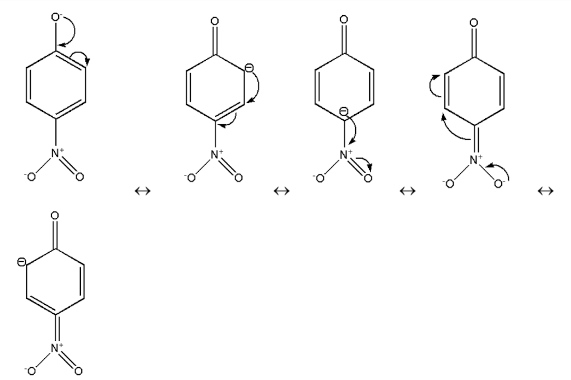 Therefore these are the resonating structures of $p - nitrophenol$.Thus, in $p - nitrophenol$ there is Stabilization due to resonance and inductive effect too.
Therefore these are the resonating structures of $p - nitrophenol$.Thus, in $p - nitrophenol$ there is Stabilization due to resonance and inductive effect too.
Hence, the correct answer is (D).
The most acidic is:
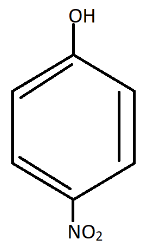
Additional information:
$N{O_2}$ Group do not show mesomeric effect of meta-position in benzene ring. Inductive effect is distance dependent. Hydrogen bonding occurs between Hydrogen, Nitrogen and Oxygen. Hydrogen bonding can be intermolecular and intramolecular.
Note:
While comparing the acidity of the different compounds, always remember the inductive effect and mesomeric effect. The stability of phenoxide ions gives us the order of acidity. More stable theoxide ${\text{ }}ion({O^ - })$, the more its acidity. The order of basicity is just the reverse of the order of acidity.
Complete answer: Let us suppose that ${H^ + }$from each compound is removed. Then, the remaining $Oxide{\text{ ion}}$ \[\left( {{O^ - }} \right)\]must be stabilized by the Nitro group.
$N{O_2}$ group has both $\left( { - I} \right)$and $\left( { - m} \right)$effect. Hence, it supports the acidic nature of compounds. With the help of $\left( { - m} \right)$effect, the conjugate base $\left( {Oxide{\text{ ion}}} \right)$gets stabilized after removal of ${H^ + }$.
Nitro is an electron withdrawing group. It will withdraw electrons of $Oxide{\text{ ion}}$\[\left( {{O^ - }} \right)\]through these two effects.
By inductive effect, it withdraws electrons, but it is an effect which is based on distance unlike mesomeric effect.

Thus, the $phenoxide{\text{ ion}}$so formed after losing the proton ${H^ + }$must be stabilized by resonance. The negative charge present on $phenoxide{\text{ ion}}$must be delocalized into a benzene ring.
Nitro group present at meta-position in$m - nitrophenol$, cannot stabilize $phenoxide{\text{ ion}}$as compared to $o - nitrophenol$ and$p - nitrophenol$.
Also, in$p - nitrophenol$, there is a chance of hydrogen bonding. (Hydrogen from $OH$ and Nitrogen from $N{O_2}$ group).

So, it is hard to lose the proton ${H^ + }$ due to $H - Bonding$ with $N{O_2}$ group.
Now, there will be more resonance effect, thus more stabilization of compound$p - nitrophenol$.

Hence, the correct answer is (D).
The most acidic is:

Additional information:
$N{O_2}$ Group do not show mesomeric effect of meta-position in benzene ring. Inductive effect is distance dependent. Hydrogen bonding occurs between Hydrogen, Nitrogen and Oxygen. Hydrogen bonding can be intermolecular and intramolecular.
Note:
While comparing the acidity of the different compounds, always remember the inductive effect and mesomeric effect. The stability of phenoxide ions gives us the order of acidity. More stable theoxide ${\text{ }}ion({O^ - })$, the more its acidity. The order of basicity is just the reverse of the order of acidity.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Ribosomal RNA is actively synthesised in A Nucleoplasm class 12 biology CBSE

How many molecules of ATP and NADPH are required information class 12 biology CBSE




