Which compound does not give HVZ reaction?
A. ${ CH }_{ 3 }-{ CH }_{ 2 }-{ CH }_{ 2 }-{ COOH }$
B.
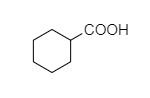
C.
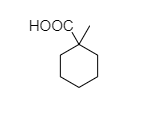
D.
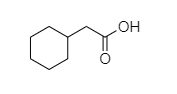



Answer
537.2k+ views
Hint: HVZ stands for Hell-Volhard-Zelinsky halogenation reaction. The HVZ reaction is halogenation of carboxylic acids at alpha carbon.
Complete answer:
This reaction is generally carried out by treating carboxylic acids with halogen in the presence of red phosphorus and then works up with water.
Those compounds which have alpha hydrogen show HVZ reaction while those which do not have alpha hydrogen do not show HVZ reaction.
A. In this ${ CH }_{ 3 }-{ CH }_{ 2 }-{ CH }_{ 2 }-{ COOH }$ compound, alpha and beta hydrogen are present. That’s why butanoic acid shows a Hell-Volhard-Zelinsky halogenation reaction.
B. In cyclohexyl carboxylic acid, alpha hydrogen is present as shown below,
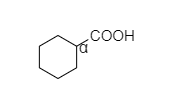
So, this compound also shows the Hell-Volhard-Zelinsky halogenation reaction.
C. In 1-methylcyclohexane-1-carboxylic acid, there is no alpha hydrogen present as the carbon is attached to 4 different bonds. So, this compound does not show the Hell-Volhard-Zelinsky halogenation reaction.
D. In 2-cyclohexyl acetic acid, alpha hydrogen is present as shown in the figure;
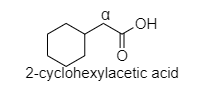
Hence, this compound shows the HVZ reaction.
The correct option is C.
Note: The possibility to make a mistake is that this reaction is specific for the replacement of alpha-hydrogens and not beta- hydrogens.
Complete answer:
This reaction is generally carried out by treating carboxylic acids with halogen in the presence of red phosphorus and then works up with water.
Those compounds which have alpha hydrogen show HVZ reaction while those which do not have alpha hydrogen do not show HVZ reaction.
A. In this ${ CH }_{ 3 }-{ CH }_{ 2 }-{ CH }_{ 2 }-{ COOH }$ compound, alpha and beta hydrogen are present. That’s why butanoic acid shows a Hell-Volhard-Zelinsky halogenation reaction.
B. In cyclohexyl carboxylic acid, alpha hydrogen is present as shown below,

So, this compound also shows the Hell-Volhard-Zelinsky halogenation reaction.
C. In 1-methylcyclohexane-1-carboxylic acid, there is no alpha hydrogen present as the carbon is attached to 4 different bonds. So, this compound does not show the Hell-Volhard-Zelinsky halogenation reaction.
D. In 2-cyclohexyl acetic acid, alpha hydrogen is present as shown in the figure;

Hence, this compound shows the HVZ reaction.
The correct option is C.
Note: The possibility to make a mistake is that this reaction is specific for the replacement of alpha-hydrogens and not beta- hydrogens.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Which among the following are examples of coming together class 11 social science CBSE




