What is the name of \[HBr\] (in water)?
Answer
520.8k+ views
Hint: Hydrogen bromide comes under the category of inorganic compound which is capable of forming hydrogen bonds due to the presence of electronegative bromine atoms. Hydrogen bromides exist in a gaseous state in the atmosphere.
Complete answer:
Hydrogen bromide is also known as hydrogen halide because halogen atoms are combined with hydrogen atoms to form hydrogen halides. Like other hydrogen halides of the halogen family, hydrogen bromide is a colorless gas which is stable in the atmosphere.
From the structure of hydrogen bromide, it is clear that hydrogen bromide molecules are capable of forming hydrogen bonds. When we dissolve hydrogen bromide in water, it forms hydrogen bonding with oxygen of water to form a solution.
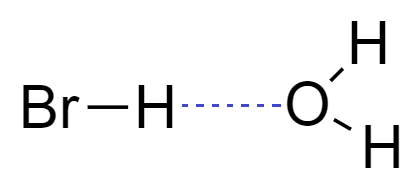
When hydrogen bromide gas dissolves into water to form a solution of Hydrogen bromide it is commonly known as hydrobromic acid.
The chemical reaction of formation of hydrobromic acid from hydrogen bromide is expressed as:
$HB{r_{\left( {s,liq,gas} \right)}} \to HB{r_{\left( {aq} \right)}}$
Aqueous hydrogen bromide is known as acid because after dissolution into water, hydrobromic acid acts as a very strong acid by releasing bromide ion and hydronium ion in the water.
Chemical reaction which shows formation of bromide ion and hydronium ion in water by hydrobromic acid is expressed as:
$HB{r_{\left( {aq} \right)}} + {H_2}O \to {H_3}{O^ + } + B{r^ - }$
Solid gases, or liquid physical state of $HBr$, is known as hydrogen bromide but aqueous $HB{r_{\left( {aq} \right)}}$ is known as hydrobromic acid.
$ \therefore $ The name of \[HBr\] (in water) is Hydrobromic acid.
Note:
Hydrobromic acid forms a saturated solution which contains $68.85\% $ of $HBr$ in it. However, hydrobromic acid that contains $HBr$ by $47.6\% $ of mass forms an azeotropic mixture which shows a boiling point near ${124.3^ \circ }C$.
Complete answer:
Hydrogen bromide is also known as hydrogen halide because halogen atoms are combined with hydrogen atoms to form hydrogen halides. Like other hydrogen halides of the halogen family, hydrogen bromide is a colorless gas which is stable in the atmosphere.
From the structure of hydrogen bromide, it is clear that hydrogen bromide molecules are capable of forming hydrogen bonds. When we dissolve hydrogen bromide in water, it forms hydrogen bonding with oxygen of water to form a solution.

When hydrogen bromide gas dissolves into water to form a solution of Hydrogen bromide it is commonly known as hydrobromic acid.
The chemical reaction of formation of hydrobromic acid from hydrogen bromide is expressed as:
$HB{r_{\left( {s,liq,gas} \right)}} \to HB{r_{\left( {aq} \right)}}$
Aqueous hydrogen bromide is known as acid because after dissolution into water, hydrobromic acid acts as a very strong acid by releasing bromide ion and hydronium ion in the water.
Chemical reaction which shows formation of bromide ion and hydronium ion in water by hydrobromic acid is expressed as:
$HB{r_{\left( {aq} \right)}} + {H_2}O \to {H_3}{O^ + } + B{r^ - }$
Solid gases, or liquid physical state of $HBr$, is known as hydrogen bromide but aqueous $HB{r_{\left( {aq} \right)}}$ is known as hydrobromic acid.
$ \therefore $ The name of \[HBr\] (in water) is Hydrobromic acid.
Note:
Hydrobromic acid forms a saturated solution which contains $68.85\% $ of $HBr$ in it. However, hydrobromic acid that contains $HBr$ by $47.6\% $ of mass forms an azeotropic mixture which shows a boiling point near ${124.3^ \circ }C$.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




