What is $P{H_3}$?
Answer
532.5k+ views
Hint: Phosphine or hydrogen phosphide is a flammable, colourless, extremely toxic gas having a disagreeable garlic-like odour. Moreover, phosphine is classed as pictogen hydride. Phosphine is usually used in semiconductor or plastic industries and even in the production of flame retardants.
Complete answer:
Phosphine is the compound with the chemical formula $P{H_3}$.
The IUPAC name for $P{H_3}$ is phosphine. Phosphine is also known as hydrogen phosphide.
Phosphine was first obtained in 1783 by Philippe Gengembre, by heating white phosphorus in an aqueous solution of potassium carbonate.
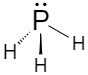
Phosphine has a trigonal pyramidal structure. Length of the P-H bond is $1.42{A^{^\circ }}$ and the H-P-H bond angle is ${93.5^ \circ }$.
Around the phosphorus atom, there are three sigma bonds and one lone pair.
Explanation of not well defined or absence of $P{H_3}$ hybridization can be given by Drago’s rule. During the formation of phosphine, the pure p orbitals participate in bonding and avoid getting hybridized.
Phosphine burns to produce a dense white cloud of phosphoric acid.
$P{H_3} + 2{O_2} \to {H_3}P{O_4}$
Phosphine dissolves less readily in water but more readily in non-polar solvents.
Phosphine is a Lewis base because of the presence of a nonbonding electron pair on the p- shell that can be donated.
Phosphine can be prepared in many ways. Industrially, it is prepared by the reaction of white phosphorus with potassium hydroxide, producing potassium hypophosphite as a by-product.
$3KOH + {P_4} + 3{H_2}O \to 3K{H_2}P{O_2} + P{H_3}$
And in the laboratory it can be prepared by disproportionation of phosphoric acid.
$4{H_3}P{O_3} \to P{H_3} + 3{H_3}P{O_4}$
Note:
Phosphine has an unpleasant odour like rotting fish and it is a highly toxic respiratory poison. Phosphine is mainly a redox toxin, which can cause cell damage by inducing oxidative stress and mitochondrial dysfunction, while it produces resistance in pests by a mutation in the mitochondrial metabolic gene.
Complete answer:
Phosphine is the compound with the chemical formula $P{H_3}$.
The IUPAC name for $P{H_3}$ is phosphine. Phosphine is also known as hydrogen phosphide.
Phosphine was first obtained in 1783 by Philippe Gengembre, by heating white phosphorus in an aqueous solution of potassium carbonate.
Phosphine has a trigonal pyramidal structure. Length of the P-H bond is $1.42{A^{^\circ }}$ and the H-P-H bond angle is ${93.5^ \circ }$.
Around the phosphorus atom, there are three sigma bonds and one lone pair.
Explanation of not well defined or absence of $P{H_3}$ hybridization can be given by Drago’s rule. During the formation of phosphine, the pure p orbitals participate in bonding and avoid getting hybridized.

Phosphine burns to produce a dense white cloud of phosphoric acid.
$P{H_3} + 2{O_2} \to {H_3}P{O_4}$
Phosphine dissolves less readily in water but more readily in non-polar solvents.
Phosphine is a Lewis base because of the presence of a nonbonding electron pair on the p- shell that can be donated.
Phosphine can be prepared in many ways. Industrially, it is prepared by the reaction of white phosphorus with potassium hydroxide, producing potassium hypophosphite as a by-product.
$3KOH + {P_4} + 3{H_2}O \to 3K{H_2}P{O_2} + P{H_3}$
And in the laboratory it can be prepared by disproportionation of phosphoric acid.
$4{H_3}P{O_3} \to P{H_3} + 3{H_3}P{O_4}$
Note:
Phosphine has an unpleasant odour like rotting fish and it is a highly toxic respiratory poison. Phosphine is mainly a redox toxin, which can cause cell damage by inducing oxidative stress and mitochondrial dysfunction, while it produces resistance in pests by a mutation in the mitochondrial metabolic gene.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Explain zero factorial class 11 maths CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




