What is distillation?
Answer
609k+ views
Hint: A lot of techniques are there to separate mixtures of compounds into individual compounds. Out of those techniques distillation is one of the methods to separate the mixture of two different liquid components.
Complete Solution :
- Distillation is a method to separate a mixture of liquid components into individual liquid components.
- The separation of liquids is based on the boiling points of the liquid components.
- Distillation is not a chemical reaction.
- Distillation process is a physical phenomenon because we are going to separate the two or more different liquids physically from one another based on their boiling point.
- First we will take the mixture of liquid components in a round bottom flask.
- Later we are going to start heating the liquid mixture.
- After a certain time the liquid mixture is going to boil, at this time the liquid component which has a low boiling point comes out first and the remaining liquid components come out basing on their boiling points.
- The liquid components which are coming out the distillation flask are going to be condensed by using a condenser and we can see it in the below picture.
- The setup for the distillation process is as follows :
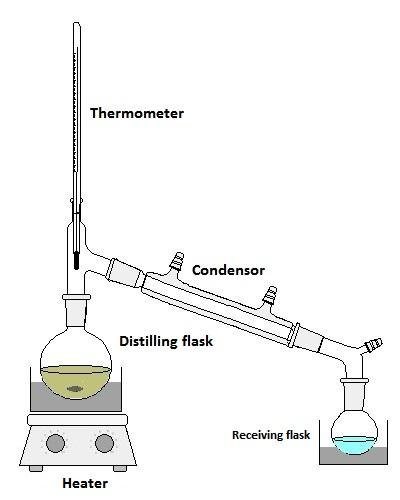
- If the liquid components have the same boiling points, we can separate them by using a fractional distillation process.
Note: There are different types of distillation processes available. These different types of distillation process are going to be used on the bases of boiling points and the presence of different impurities in the liquid mixtures.
Complete Solution :
- Distillation is a method to separate a mixture of liquid components into individual liquid components.
- The separation of liquids is based on the boiling points of the liquid components.
- Distillation is not a chemical reaction.
- Distillation process is a physical phenomenon because we are going to separate the two or more different liquids physically from one another based on their boiling point.
- First we will take the mixture of liquid components in a round bottom flask.
- Later we are going to start heating the liquid mixture.
- After a certain time the liquid mixture is going to boil, at this time the liquid component which has a low boiling point comes out first and the remaining liquid components come out basing on their boiling points.
- The liquid components which are coming out the distillation flask are going to be condensed by using a condenser and we can see it in the below picture.
- The setup for the distillation process is as follows :

- If the liquid components have the same boiling points, we can separate them by using a fractional distillation process.
Note: There are different types of distillation processes available. These different types of distillation process are going to be used on the bases of boiling points and the presence of different impurities in the liquid mixtures.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
Find the sum of series 1 + 2 + 3 + 4 + 5 + + 100 class 9 maths CBSE

Difference Between Plant Cell and Animal Cell

Fill the blanks with the suitable prepositions 1 The class 9 english CBSE

What is pollution? How many types of pollution? Define it

What is the Full Form of ICSE / ISC ?

Name 10 Living and Non living things class 9 biology CBSE




