
How many total isomers for $ \left[ Co{{\left( en \right)}_{2}}C{{l}_{2}} \right]N{{O}_{2}} $ ?
Answer
519k+ views
Hint: We know that the given complex has stereoisomers and it does not have structural isomers. One of the isomers of this complex can rotate the plane of the polarized light. Configurational isomers are the stereoisomers that cannot be converted into one another by rotation around a single bond in the molecule.
Complete answer:
Isomers are the species which have the same molecular formula but different properties. Isomers can be divided into two main categories: structural isomers and stereoisomers. Structural isomerism can also be divided into categories like linkage isomerism, coordination isomerism, ionization isomerism and solvate isomerism. There are two types of Configurational isomers:
-Geometrical Isomers
-Optical Isomers.
Geometrical isomerism is going to show by unsaturated compounds and optical isomerism is going to exhibit by the molecules that are having chiral carbons.
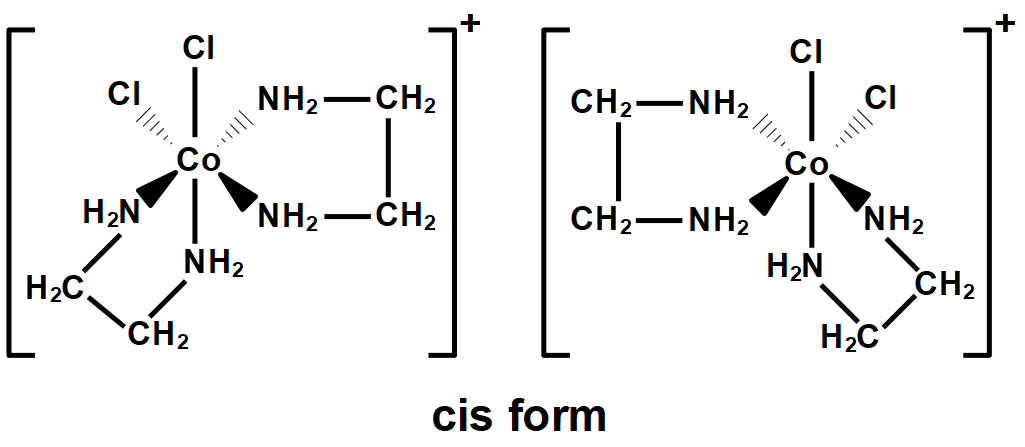
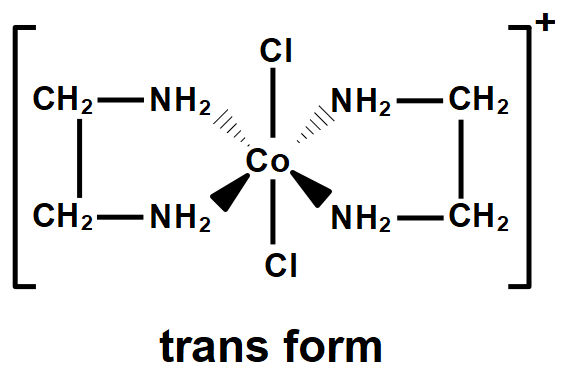
The given compound will have two geometric isomers; cis and trans. Optical isomers are the mirror images of the compounds that cannot be superimposed on each other. So, the cis isomer can form such two isomers (including itself) that are enantiomers. The optical isomers are named as dextro and laevo depending upon its characteristic to rotate the plane polarized light.
Therefore, the total isomers for $ \left[ Co{{\left( en \right)}_{2}}C{{l}_{2}} \right]N{{O}_{2}} $ are $ 3. $
Note:
Remember that don’t be confused between geometrical isomerism and optical isomerism. The compounds that have double bonds will show geometrical isomerism and the compounds which have chiral centers show the optical isomerism.
Complete answer:
Isomers are the species which have the same molecular formula but different properties. Isomers can be divided into two main categories: structural isomers and stereoisomers. Structural isomerism can also be divided into categories like linkage isomerism, coordination isomerism, ionization isomerism and solvate isomerism. There are two types of Configurational isomers:
-Geometrical Isomers
-Optical Isomers.
Geometrical isomerism is going to show by unsaturated compounds and optical isomerism is going to exhibit by the molecules that are having chiral carbons.


The given compound will have two geometric isomers; cis and trans. Optical isomers are the mirror images of the compounds that cannot be superimposed on each other. So, the cis isomer can form such two isomers (including itself) that are enantiomers. The optical isomers are named as dextro and laevo depending upon its characteristic to rotate the plane polarized light.
Therefore, the total isomers for $ \left[ Co{{\left( en \right)}_{2}}C{{l}_{2}} \right]N{{O}_{2}} $ are $ 3. $
Note:
Remember that don’t be confused between geometrical isomerism and optical isomerism. The compounds that have double bonds will show geometrical isomerism and the compounds which have chiral centers show the optical isomerism.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

The largest wind power cluster is located in the state class 11 social science CBSE

Explain zero factorial class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Which among the following are examples of coming together class 11 social science CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE




