To prepare propan-2-ol from methyl magnesium iodide, the chemical reagent required is:
A.\[C{H_3}CHO\]
B.\[HCHO\]
C.\[C{H_3}COC{H_3}\]
D.\[C{O_2}\]
Answer
545.1k+ views
Hint: We must have to know that the propan-2-ol, otherwise known as isopropyl alcohol, is a chemical compound having the formula \[{C_3}{H_8}O\]. Here the isopropyl group is attached with a hydroxyl group and there is a formation of secondary alcohol. And the alcohol carbon atom is linked with two different carbon atoms. The propan – 2- ol is mainly used for the preparation of solvents like dye solutions, soap, etc.
Complete answer:
When the acetaldehyde is reacting with methyl magnesium iodide, there is a formation of secondary alcohol which is propan – 2 – ol.
Here the methyl magnesium iodide is linked with carbonyl carbon and there is a formation of adduct. And due to the electronegativity difference, adducts have partial charges. And thus adduct is reacted with water and undergoes the hydrolysis, will get propan – 2 – ol.
Let’s see the reaction,
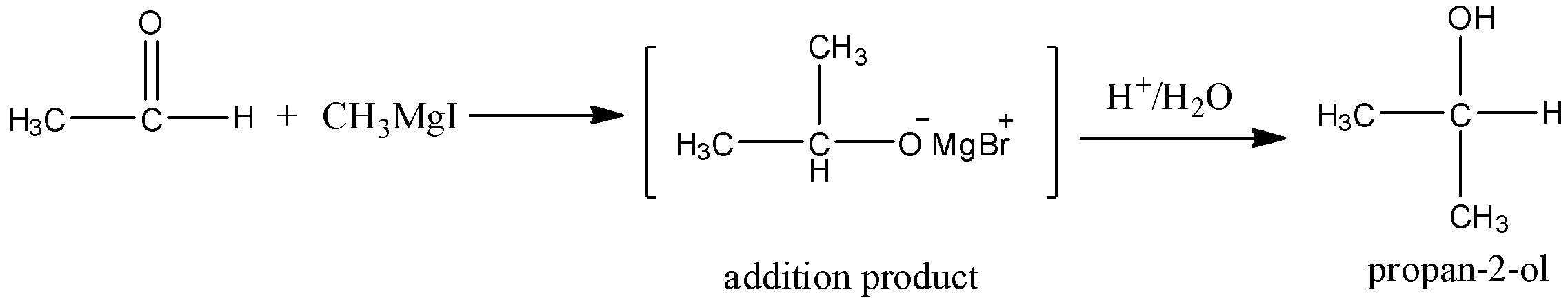
Hence, option (A) is correct.
When the formaldehyde is reacting with methyl magnesium iodide, (Grignard reagent), there is a formation of primary alcohol. But here, propan -2- ol is a secondary alcohol. Hence, the option (B) is incorrect.
When acetone is reacting with methyl magnesium iodide, there is a formation of 2 -methyl 2 propanol and will not get propan – 2 – ol. Hence, option (C) is incorrect.
When the carbon dioxide is reacting with methyl magnesium bromide, the product should be a carboxylic acid and not give propan – 2 – ol. Hence, the option (D) is incorrect.
Hence, option (A) is correct.
Note:
We need to know that the Grignard reagent is a chemical compound which is widely used as a reagent in organic synthesis. The general formula of Grignard reagent is, \[R - Mg - X\]. If the Grignard reagent is reacted with aldehyde or ketone, there is a formation of secondary alcohol and tertiary alcohol respectively. But, when the formaldehyde is reacting with Grignard reagent, there is a formation of primary alcohol.
Complete answer:
When the acetaldehyde is reacting with methyl magnesium iodide, there is a formation of secondary alcohol which is propan – 2 – ol.
Here the methyl magnesium iodide is linked with carbonyl carbon and there is a formation of adduct. And due to the electronegativity difference, adducts have partial charges. And thus adduct is reacted with water and undergoes the hydrolysis, will get propan – 2 – ol.
Let’s see the reaction,

Hence, option (A) is correct.
When the formaldehyde is reacting with methyl magnesium iodide, (Grignard reagent), there is a formation of primary alcohol. But here, propan -2- ol is a secondary alcohol. Hence, the option (B) is incorrect.
When acetone is reacting with methyl magnesium iodide, there is a formation of 2 -methyl 2 propanol and will not get propan – 2 – ol. Hence, option (C) is incorrect.
When the carbon dioxide is reacting with methyl magnesium bromide, the product should be a carboxylic acid and not give propan – 2 – ol. Hence, the option (D) is incorrect.
Hence, option (A) is correct.
Note:
We need to know that the Grignard reagent is a chemical compound which is widely used as a reagent in organic synthesis. The general formula of Grignard reagent is, \[R - Mg - X\]. If the Grignard reagent is reacted with aldehyde or ketone, there is a formation of secondary alcohol and tertiary alcohol respectively. But, when the formaldehyde is reacting with Grignard reagent, there is a formation of primary alcohol.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
Difference Between Prokaryotic Cells and Eukaryotic Cells

Draw a diagram of nephron and explain its structur class 11 biology CBSE

Net gain of ATP in glycolysis a 6 b 2 c 4 d 8 class 11 biology CBSE

Difference between physical and chemical change class 11 chemistry CBSE

Which of the following is the hottest month AMay BJune class 11 social science CBSE

How do I convert ms to kmh Give an example class 11 physics CBSE




