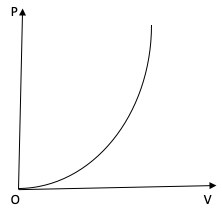
The variation of pressure P with volume V for an ideal diatomic gas is parabolic as shown in the figure. The molar specific heat of the gas during this process is?
A.$\dfrac{{9R}}{5}$
B.$\dfrac{{17R}}{6}$
C.$\dfrac{{3R}}{5}$
D.$\dfrac{{8R}}{5}$

Answer
598.5k+ views
Hint: From the PV curve given in the question, we will find the relation between P and V, and then by comparing it with the relation for the polytropic process $(P{V^x} = const.)$ we can find the value of ‘x’. Then by using the below given formula, molar specific heat of the polytropic process can be found.
$C = \dfrac{{fR}}{2} + \dfrac{R}{{1 - x}}$
Complete step by step answer:
The PV curve of the diatomic gas is a parabola. Therefore, we can write
$P\alpha {V^2}$
$ \Rightarrow P{V^{ - 2}} = $ constant
For a polytropic process, the relation between P and V is $P{V^x} = $ constant. On comparing the two equations we get, $x = - 2$.
Now, in case of a diatomic gas, the degree of freedom is 5, that is, $f = 5$.
The molar specific heat of a gas undergoing polytropic process is given by,
$C = \dfrac{{fR}}{2} + \dfrac{R}{{1 - x}}$
Substituting the values of ‘f’ and ‘x’, we get,
$C = \dfrac{{5R}}{2} + \dfrac{R}{{1 - ( - 2)}}$
$ \Rightarrow C = \dfrac{{5R}}{2} + \dfrac{R}{{1 + 2}}$
$ \Rightarrow C = \dfrac{{5R}}{2} + \dfrac{R}{3}$
$ \Rightarrow C = \dfrac{{15R + 2R}}{6}$
$ \Rightarrow C = \dfrac{{17R}}{6}$
Thus, the molar specific heat of the given diatomic gas is $\dfrac{{17R}}{6}$.
Hence option B is correct.
Note:
The molar specific heat of gases is the amount of heat energy required by 1 mole of the gas to raise its temperature by 1 degree Celsius. There are two types of molar specific heats of gases. When measured at constant pressure, it is called the molar specific heat at constant pressure, denoted by ${C_p}$. When it is measured at constant volume, then it is called molar specific heat at constant volume, denoted by ${C_v}$. Both these quantities are related as ${C_p} = {C_v} + R$.
$C = \dfrac{{fR}}{2} + \dfrac{R}{{1 - x}}$
Complete step by step answer:
The PV curve of the diatomic gas is a parabola. Therefore, we can write
$P\alpha {V^2}$
$ \Rightarrow P{V^{ - 2}} = $ constant
For a polytropic process, the relation between P and V is $P{V^x} = $ constant. On comparing the two equations we get, $x = - 2$.
Now, in case of a diatomic gas, the degree of freedom is 5, that is, $f = 5$.
The molar specific heat of a gas undergoing polytropic process is given by,
$C = \dfrac{{fR}}{2} + \dfrac{R}{{1 - x}}$
Substituting the values of ‘f’ and ‘x’, we get,
$C = \dfrac{{5R}}{2} + \dfrac{R}{{1 - ( - 2)}}$
$ \Rightarrow C = \dfrac{{5R}}{2} + \dfrac{R}{{1 + 2}}$
$ \Rightarrow C = \dfrac{{5R}}{2} + \dfrac{R}{3}$
$ \Rightarrow C = \dfrac{{15R + 2R}}{6}$
$ \Rightarrow C = \dfrac{{17R}}{6}$
Thus, the molar specific heat of the given diatomic gas is $\dfrac{{17R}}{6}$.
Hence option B is correct.
Note:
The molar specific heat of gases is the amount of heat energy required by 1 mole of the gas to raise its temperature by 1 degree Celsius. There are two types of molar specific heats of gases. When measured at constant pressure, it is called the molar specific heat at constant pressure, denoted by ${C_p}$. When it is measured at constant volume, then it is called molar specific heat at constant volume, denoted by ${C_v}$. Both these quantities are related as ${C_p} = {C_v} + R$.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Which among the following are examples of coming together class 11 social science CBSE




