The terminal and bridged ${\text{CO}}$ ligands in the compound $\left[ {{\text{C}}{{\text{o}}_{\text{2}}}{{\left( {{\text{CO}}} \right)}_{\text{8}}}} \right]$ are respectively:
A. ${\text{0,2}}$
B. ${\text{6,1}}$
C. ${\text{5,2}}$
D. ${\text{6,2}}$
Answer
609.6k+ views
Hint: Calculate the total electrons for the complex. Place the bonds in such a way that the total electron count is satisfied.
The metal atom ${\text{Co}}$ contributes nine electrons each.
The ${\text{CO}}$ ligand at terminal position donates two electrons to each metal and one electron to each metal when bridged.
Complete step by step answer:
Step 1:
Calculate the total electrons for the complex $\left[ {{\text{C}}{{\text{o}}_{\text{2}}}{{\left( {{\text{CO}}} \right)}_{\text{8}}}} \right]$.
The metal ${\text{Co}}$ contributes nine electrons each while the ligand ${\text{CO}}$ contributes two electrons each. Thus,
Total electron count for $\left[ {{\text{C}}{{\text{o}}_{\text{2}}}{{\left( {{\text{CO}}} \right)}_{\text{8}}}} \right]$ $ = \left( {2 \times {\text{Electrons of Co}}} \right) + \left( {8 \times {\text{Electrons of CO}}} \right)$
$ = \left( {2 \times 9} \right) + \left( {8 \times 2} \right)$
$ = 18 + 16$
Total electron count for $\left[ {{\text{C}}{{\text{o}}_{\text{2}}}{{\left( {{\text{CO}}} \right)}_{\text{8}}}} \right]$ $ = 34$
Step 2:
Calculate the number of metal – metal bonds per metal:
Number of metal - metal bonds per metal $ = \dfrac{{\left( {{\text{Number of metal atoms}} \times 18} \right) - {\text{Total electron count}}}}{2}$
The complex $\left[ {{\text{C}}{{\text{o}}_{\text{2}}}{{\left( {{\text{CO}}} \right)}_{\text{8}}}} \right]$ has $2$ ${\text{Co}}$metal atoms. Thus, substitute $2$ for the number of metal atoms and ${\text{34}}$ for the total electron count. Thus,
Number of metal - metal bonds per metal $ = \dfrac{{\left( {2 \times 18} \right) - {\text{34}}}}{2}$
$ = \dfrac{{36 - {\text{34}}}}{2}$
$ = \dfrac{2}{2}$
Number of metal - metal bonds per metal $ = 1$
Step 3:
Draw the structure for the complex $\left[ {{\text{C}}{{\text{o}}_{\text{2}}}{{\left( {{\text{CO}}} \right)}_{\text{8}}}} \right]$:
There is one metal – metal bond i.e. there is one bond between ${\text{Co}} - {\text{Co}}$.
Each ${\text{Co}}$ atom contributes nine electrons.
The ${\text{CO}}$ ligand at terminal position donates two electrons to each metal and one electron to each metal when bridged. Thus, the structure for the complex is,
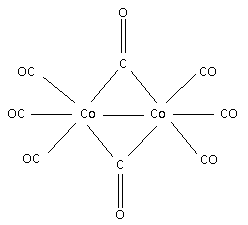
Step 4:
Calculate the number of terminal and bridged ${\text{CO}}$ ligands.
From the structure,
There are $6$ terminal ${\text{CO}}$ ligands and $2$ bridged ${\text{CO}}$ ligands.
Thus, the terminal and bridged ${\text{CO}}$ ligands in the compound $\left[ {{\text{C}}{{\text{o}}_{\text{2}}}{{\left( {{\text{CO}}} \right)}_{\text{8}}}} \right]$ are $6{\text{ and 2}}$ respectively.
Thus, the correct option is option (D).
Note: The metal atom ${\text{Co}}$ contributes nine electrons each. The ${\text{CO}}$ ligand at terminal position donates two electrons to each metal and one electron to each metal when bridged. Placing the bonds accurately to satisfy the total electron count gives the exact number of terminal and bridged ligands.
The metal atom ${\text{Co}}$ contributes nine electrons each.
The ${\text{CO}}$ ligand at terminal position donates two electrons to each metal and one electron to each metal when bridged.
Complete step by step answer:
Step 1:
Calculate the total electrons for the complex $\left[ {{\text{C}}{{\text{o}}_{\text{2}}}{{\left( {{\text{CO}}} \right)}_{\text{8}}}} \right]$.
The metal ${\text{Co}}$ contributes nine electrons each while the ligand ${\text{CO}}$ contributes two electrons each. Thus,
Total electron count for $\left[ {{\text{C}}{{\text{o}}_{\text{2}}}{{\left( {{\text{CO}}} \right)}_{\text{8}}}} \right]$ $ = \left( {2 \times {\text{Electrons of Co}}} \right) + \left( {8 \times {\text{Electrons of CO}}} \right)$
$ = \left( {2 \times 9} \right) + \left( {8 \times 2} \right)$
$ = 18 + 16$
Total electron count for $\left[ {{\text{C}}{{\text{o}}_{\text{2}}}{{\left( {{\text{CO}}} \right)}_{\text{8}}}} \right]$ $ = 34$
Step 2:
Calculate the number of metal – metal bonds per metal:
Number of metal - metal bonds per metal $ = \dfrac{{\left( {{\text{Number of metal atoms}} \times 18} \right) - {\text{Total electron count}}}}{2}$
The complex $\left[ {{\text{C}}{{\text{o}}_{\text{2}}}{{\left( {{\text{CO}}} \right)}_{\text{8}}}} \right]$ has $2$ ${\text{Co}}$metal atoms. Thus, substitute $2$ for the number of metal atoms and ${\text{34}}$ for the total electron count. Thus,
Number of metal - metal bonds per metal $ = \dfrac{{\left( {2 \times 18} \right) - {\text{34}}}}{2}$
$ = \dfrac{{36 - {\text{34}}}}{2}$
$ = \dfrac{2}{2}$
Number of metal - metal bonds per metal $ = 1$
Step 3:
Draw the structure for the complex $\left[ {{\text{C}}{{\text{o}}_{\text{2}}}{{\left( {{\text{CO}}} \right)}_{\text{8}}}} \right]$:
There is one metal – metal bond i.e. there is one bond between ${\text{Co}} - {\text{Co}}$.
Each ${\text{Co}}$ atom contributes nine electrons.
The ${\text{CO}}$ ligand at terminal position donates two electrons to each metal and one electron to each metal when bridged. Thus, the structure for the complex is,

Step 4:
Calculate the number of terminal and bridged ${\text{CO}}$ ligands.
From the structure,
There are $6$ terminal ${\text{CO}}$ ligands and $2$ bridged ${\text{CO}}$ ligands.
Thus, the terminal and bridged ${\text{CO}}$ ligands in the compound $\left[ {{\text{C}}{{\text{o}}_{\text{2}}}{{\left( {{\text{CO}}} \right)}_{\text{8}}}} \right]$ are $6{\text{ and 2}}$ respectively.
Thus, the correct option is option (D).
Note: The metal atom ${\text{Co}}$ contributes nine electrons each. The ${\text{CO}}$ ligand at terminal position donates two electrons to each metal and one electron to each metal when bridged. Placing the bonds accurately to satisfy the total electron count gives the exact number of terminal and bridged ligands.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE




