The structure of $C{{H}_{2}}=C{{H}_{2}}$ is
(A)- Linear
(B)- Planar
(C)- Non-planar
(D)- None of the above
Answer
628.5k+ views
Hint: The carbon atoms in $C{{H}_{2}}=C{{H}_{2}}$ are $s{{p}^{2}}$ hybridized. The hybridization of a molecule tells us about the geometry and shape of the molecule.
Complete step by step solution:
Let’s look at the answer
- The carbon atoms in the given molecule are both $s{{p}^{2}}$ hybridized.
-The shape of $s{{p}^{2}}$ hybridization is triangular planar.
-The carbon atom in the given molecule is linked to 2 sigma bonds to H atoms and one pi bond to another carbon atom.
-The pi bonds are formed by the lateral overlapping. For this the two atoms should be in the same plane for maximum overlap.
-In triangular planar geometry all the four atoms are in the same plane and the bond angles are$120{}^\circ $.
-The geometries of both the carbon atoms in the given molecule are triangular planar which is a planar geometry and the shape is also the same. Since, both the atoms are joined by double bonds so the molecule as a whole is planar.
Hence, the given molecule is a planar molecule.
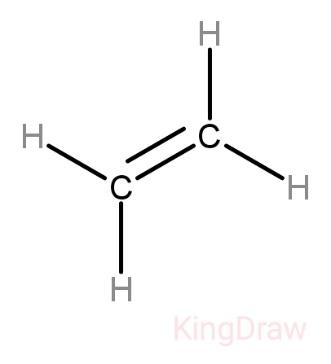
Structure of ethene
So, the correct answer is “Option B”.
Note: $C{{H}_{2}}=C{{H}_{2}}$is an alkene. In an alkene the single bond lengths and double bond lengths are different. It is also more acidic in nature than the corresponding alkane due to its geometry.
Complete step by step solution:
Let’s look at the answer
- The carbon atoms in the given molecule are both $s{{p}^{2}}$ hybridized.
-The shape of $s{{p}^{2}}$ hybridization is triangular planar.
-The carbon atom in the given molecule is linked to 2 sigma bonds to H atoms and one pi bond to another carbon atom.
-The pi bonds are formed by the lateral overlapping. For this the two atoms should be in the same plane for maximum overlap.
-In triangular planar geometry all the four atoms are in the same plane and the bond angles are$120{}^\circ $.
-The geometries of both the carbon atoms in the given molecule are triangular planar which is a planar geometry and the shape is also the same. Since, both the atoms are joined by double bonds so the molecule as a whole is planar.
Hence, the given molecule is a planar molecule.

Structure of ethene
So, the correct answer is “Option B”.
Note: $C{{H}_{2}}=C{{H}_{2}}$is an alkene. In an alkene the single bond lengths and double bond lengths are different. It is also more acidic in nature than the corresponding alkane due to its geometry.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




