The reagent used for converting benzene to ethylbenzene is:
A. \[{C_2}{H_5}CI\] , anhydrous \[AlC{l_3}\]
B. \[{C_2}{H_5}CI\] , aq \[AlC{l_3}\]
C. \[{C_2}{H_5}OH\] , anhydrous \[AlC{l_3}\]
D. \[{C_2}{H_5}CI\] , \[SOC{l_2}\]
Answer
618k+ views
Hint: Reagent can be a substance or mixture of compounds which are basically used in chemical analysis or reactions. When a reagent is added to a system it causes chemical reactions. For a particular reaction a particular reagent gets consumed in the process of the chemical reaction.
Complete step by step answer:
There are so many different kinds of reaction sequence pathways to do this conversion. The use of every reagent which is used in conversions should be known. Always find out the change of groups and make a suitable reaction sequence for conversions.
To convert benzene to ethylbenzene there is a very well-known reaction which is the Friedel-Craft reaction. It is basically a coupling reaction between aromatic compounds (benzene) and alkyl halide or acyl halide in presence of anhydrous Lewis acid \[AlC{l_3}\] . In presence of aq. \[AlC{l_3}\] it reduces the nucleophilicity of the benzene ring by formation of cation of benzene ring. The reaction is shown below,
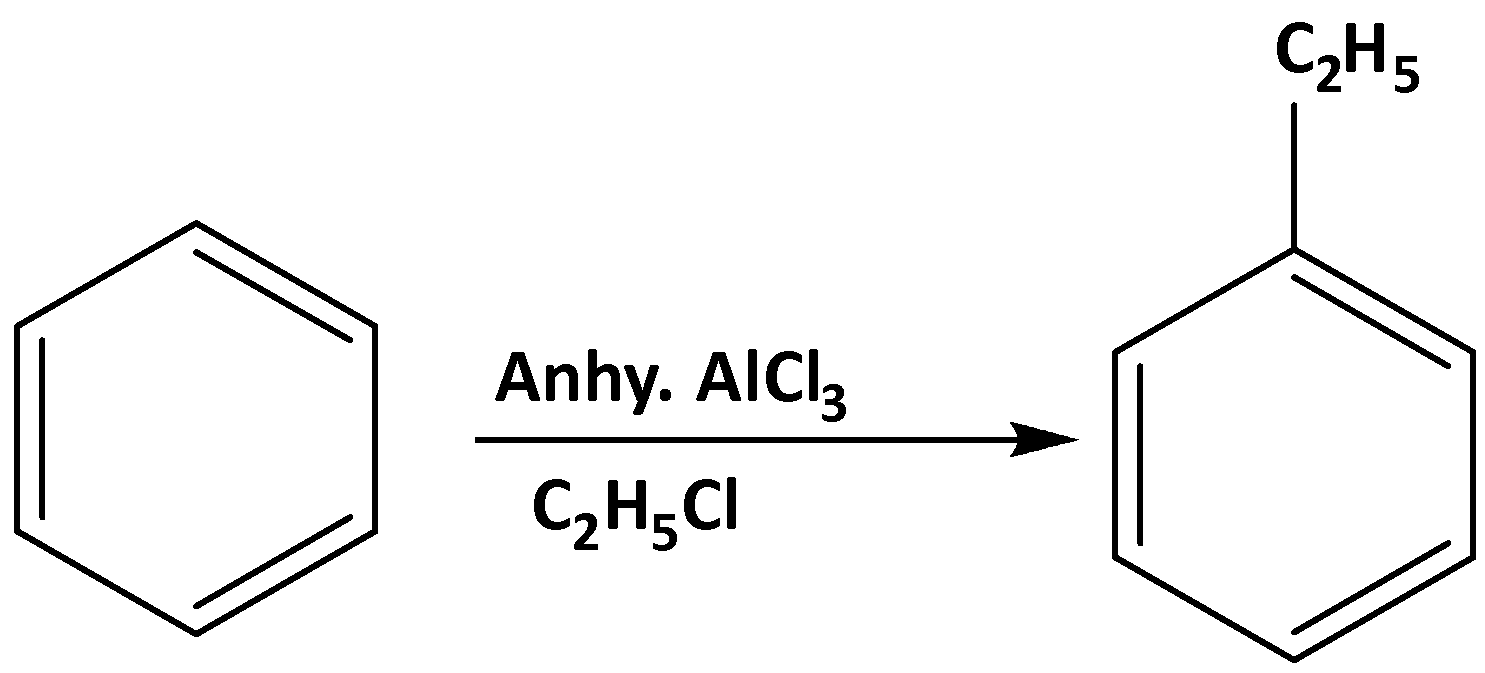
Therefore, the reagent used for converting benzene to ethylbenzene is: \[{C_2}{H_5}CI\] , anhydrous \[AlC{l_3}\]
So, the correct option is A.
Note: Friedel-Craft reaction with aryl halide is not possible because the lone pair of halogen undergoes conjugation with the benzene ring and forms a partial double bond. Due to this reason, only alkyl halide can be used as a halide component in a Friedel-craft reaction.
Complete step by step answer:
There are so many different kinds of reaction sequence pathways to do this conversion. The use of every reagent which is used in conversions should be known. Always find out the change of groups and make a suitable reaction sequence for conversions.
To convert benzene to ethylbenzene there is a very well-known reaction which is the Friedel-Craft reaction. It is basically a coupling reaction between aromatic compounds (benzene) and alkyl halide or acyl halide in presence of anhydrous Lewis acid \[AlC{l_3}\] . In presence of aq. \[AlC{l_3}\] it reduces the nucleophilicity of the benzene ring by formation of cation of benzene ring. The reaction is shown below,

Therefore, the reagent used for converting benzene to ethylbenzene is: \[{C_2}{H_5}CI\] , anhydrous \[AlC{l_3}\]
So, the correct option is A.
Note: Friedel-Craft reaction with aryl halide is not possible because the lone pair of halogen undergoes conjugation with the benzene ring and forms a partial double bond. Due to this reason, only alkyl halide can be used as a halide component in a Friedel-craft reaction.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

What are hard magnetic materials class 11 physics CBSE




