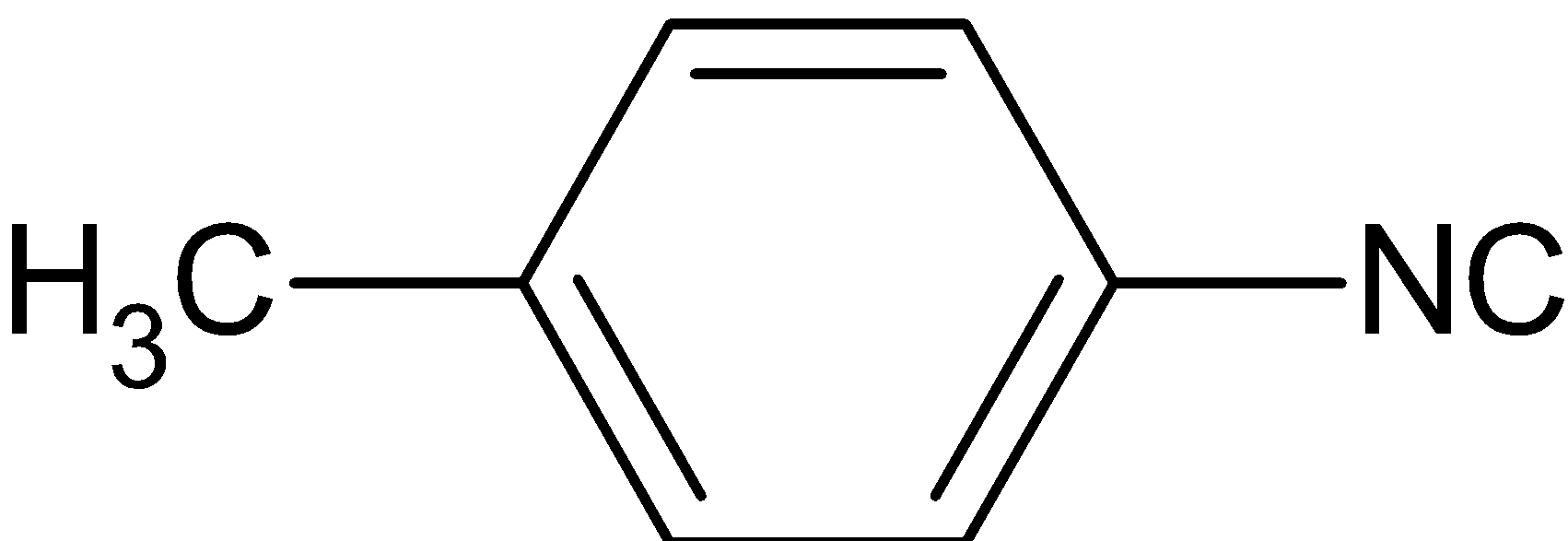
The reaction of chloroform with alcoholic ${\text{KOH}}$ and p-toluidine forms:
A.
B.
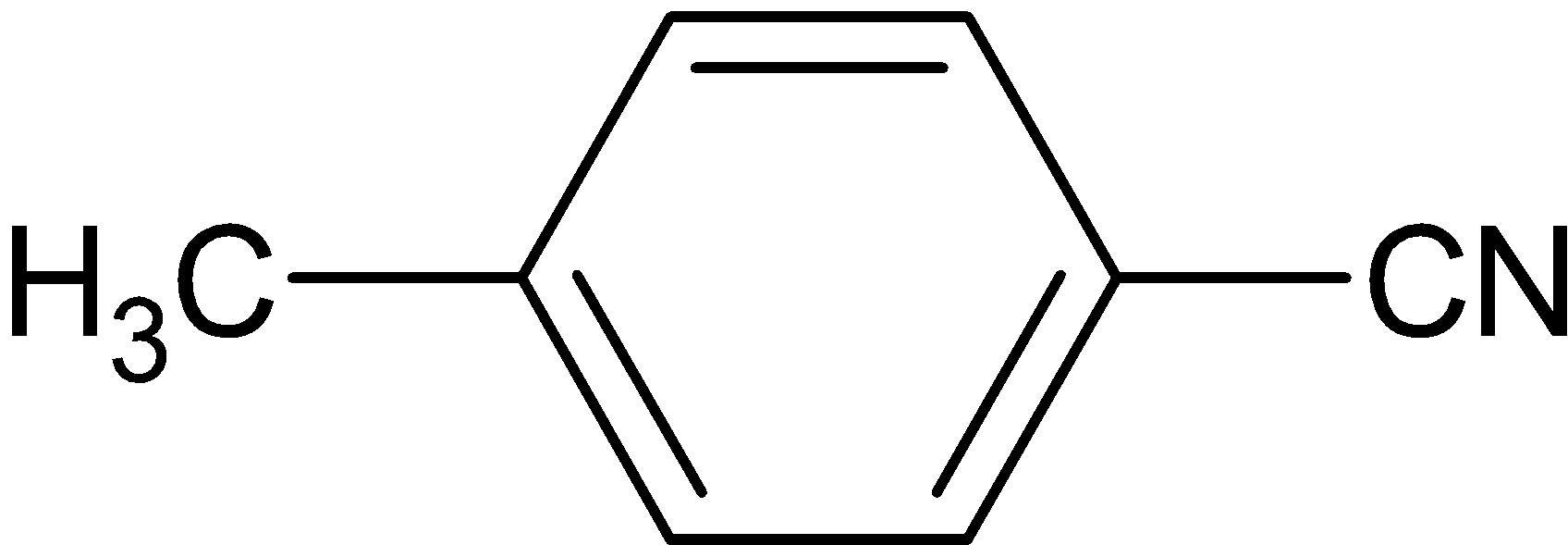
C.
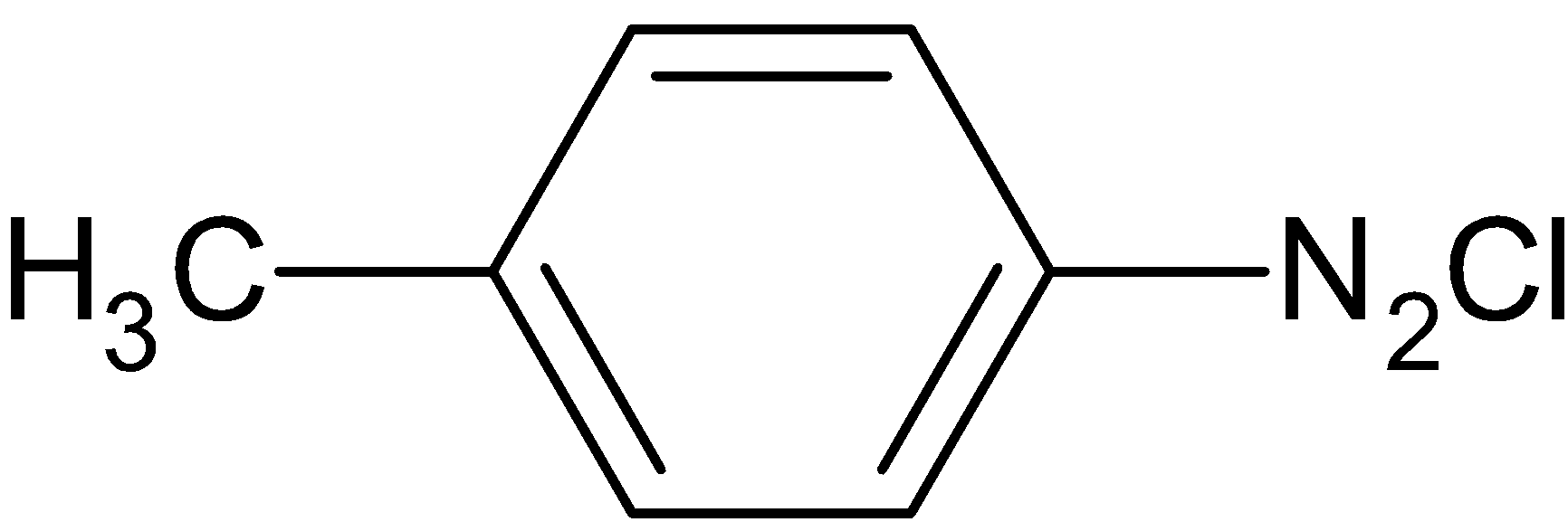
D.
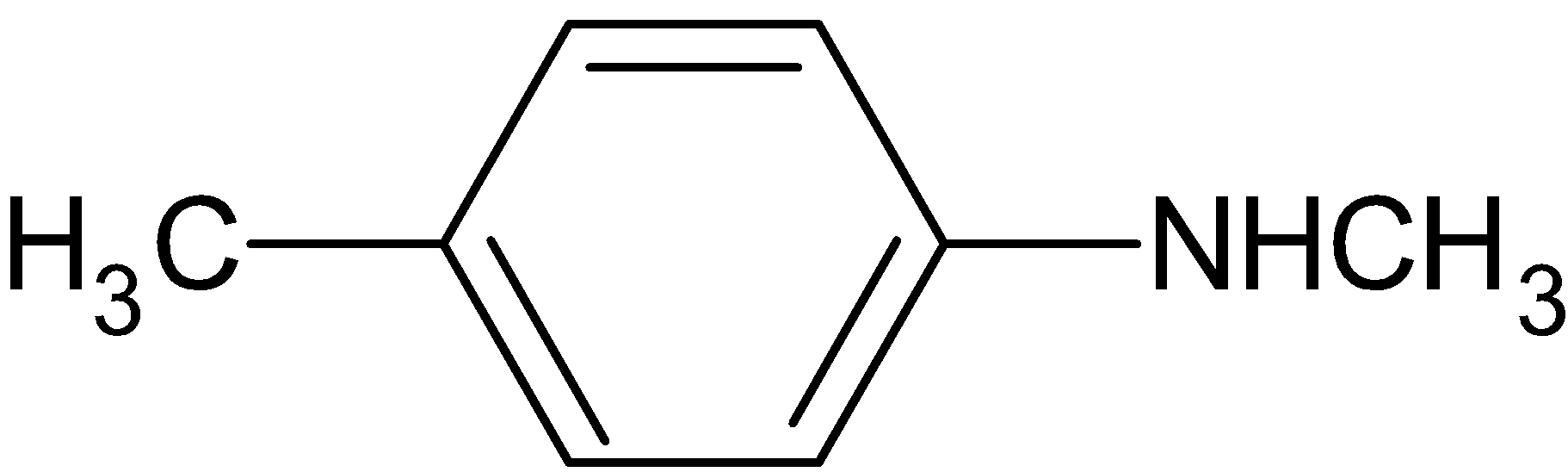




Answer
598.2k+ views
Hint:Chloroform and alcoholic potassium hydroxide are used to carry out the carbylamine reaction. It is a reaction shown only by primary amines, both aliphatic and aromatic. The attacking group is a carbene.
Complete answer:
Chloroform is tri- chloro methane and is given by the formula $CHC{l_3}$. The chemical structure of para- toluidine can be drawn as
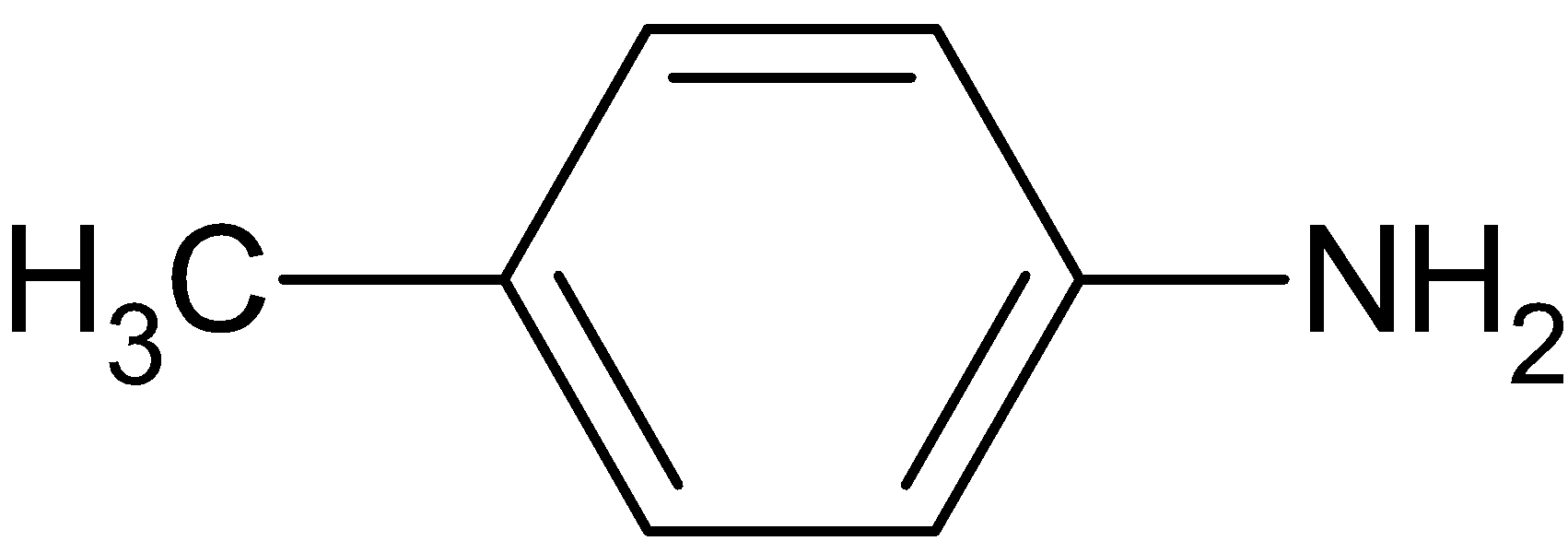
The reaction of primary amines with chloroform in the presence of alcoholic solution of an alkali is known as Hofmann isocyanide synthesis of carbylamine reaction. We know that alcoholic solution of ${\text{KOH}}$ causes dehydrohalogenation in alkyl halides and thus chloroform is converted into dichlorocarbene. Dichlorocarbene is a neutral but electron deficient species. The electrophilic carbene group attacks the nucleophilic nitrogen of the amine. Further dehydrohalogenation of the compound formed yields isocyanide.
The product formed is
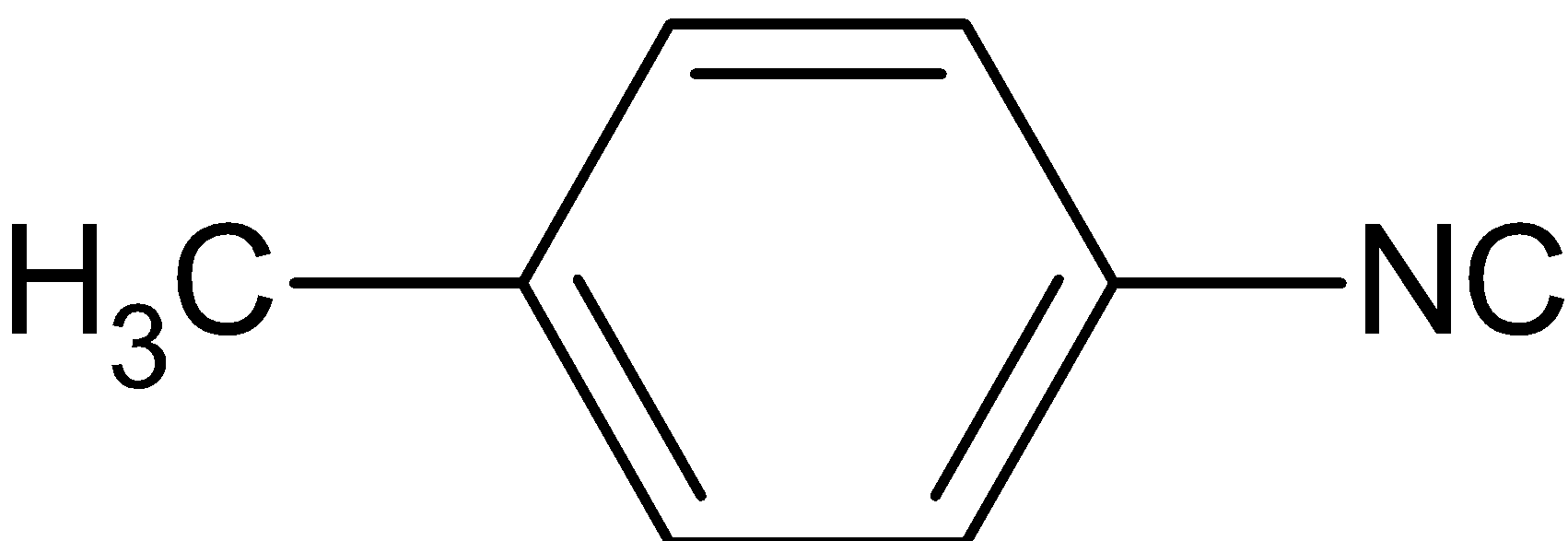
Thus, the correct answer is A.
Additional Information:
This reaction is shown only by primary amines and not by secondary or tertiary amines. Hence, this reaction can be used to detect the presence of primary amines. For the test, the amine is heated in the test tube with chloroform and alcoholic potassium hydroxide. If the compound contains primary amine, isocyanide is formed thus, imparting a foul smell.
Note:
When we use an alcoholic solution of ${\text{KOH}}$, the attacking species formed is different. In alcoholic solution ${\text{KOH}}$ forms an alkoxide ion, which is a strong base. The alkoxide ion extracts a proton from the $\beta $- carbon atom of the alkyl chloride while the chlorine atom leaves as chloride ion. Hence, if the solution used was alcoholic ${\text{KOH}}$, then the compound would have undergone dehydrohalogenation reaction twice forming an alkyne.
Complete answer:
Chloroform is tri- chloro methane and is given by the formula $CHC{l_3}$. The chemical structure of para- toluidine can be drawn as

The reaction of primary amines with chloroform in the presence of alcoholic solution of an alkali is known as Hofmann isocyanide synthesis of carbylamine reaction. We know that alcoholic solution of ${\text{KOH}}$ causes dehydrohalogenation in alkyl halides and thus chloroform is converted into dichlorocarbene. Dichlorocarbene is a neutral but electron deficient species. The electrophilic carbene group attacks the nucleophilic nitrogen of the amine. Further dehydrohalogenation of the compound formed yields isocyanide.
The product formed is

Thus, the correct answer is A.
Additional Information:
This reaction is shown only by primary amines and not by secondary or tertiary amines. Hence, this reaction can be used to detect the presence of primary amines. For the test, the amine is heated in the test tube with chloroform and alcoholic potassium hydroxide. If the compound contains primary amine, isocyanide is formed thus, imparting a foul smell.
Note:
When we use an alcoholic solution of ${\text{KOH}}$, the attacking species formed is different. In alcoholic solution ${\text{KOH}}$ forms an alkoxide ion, which is a strong base. The alkoxide ion extracts a proton from the $\beta $- carbon atom of the alkyl chloride while the chlorine atom leaves as chloride ion. Hence, if the solution used was alcoholic ${\text{KOH}}$, then the compound would have undergone dehydrohalogenation reaction twice forming an alkyne.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




