The product formed should be:
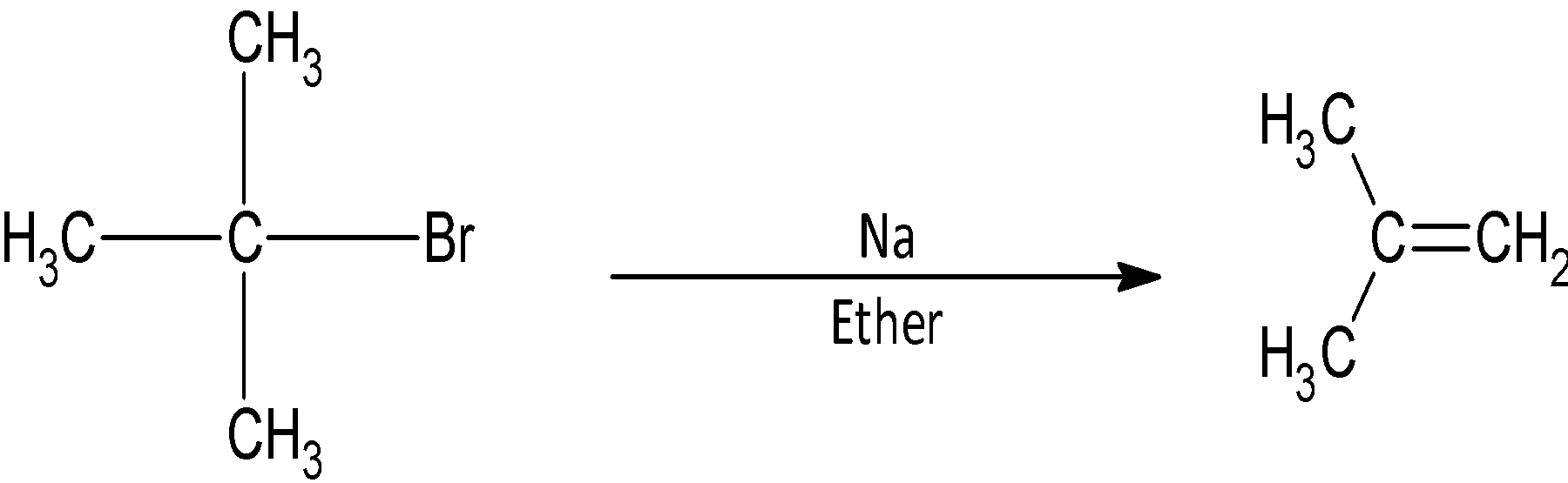
(A)
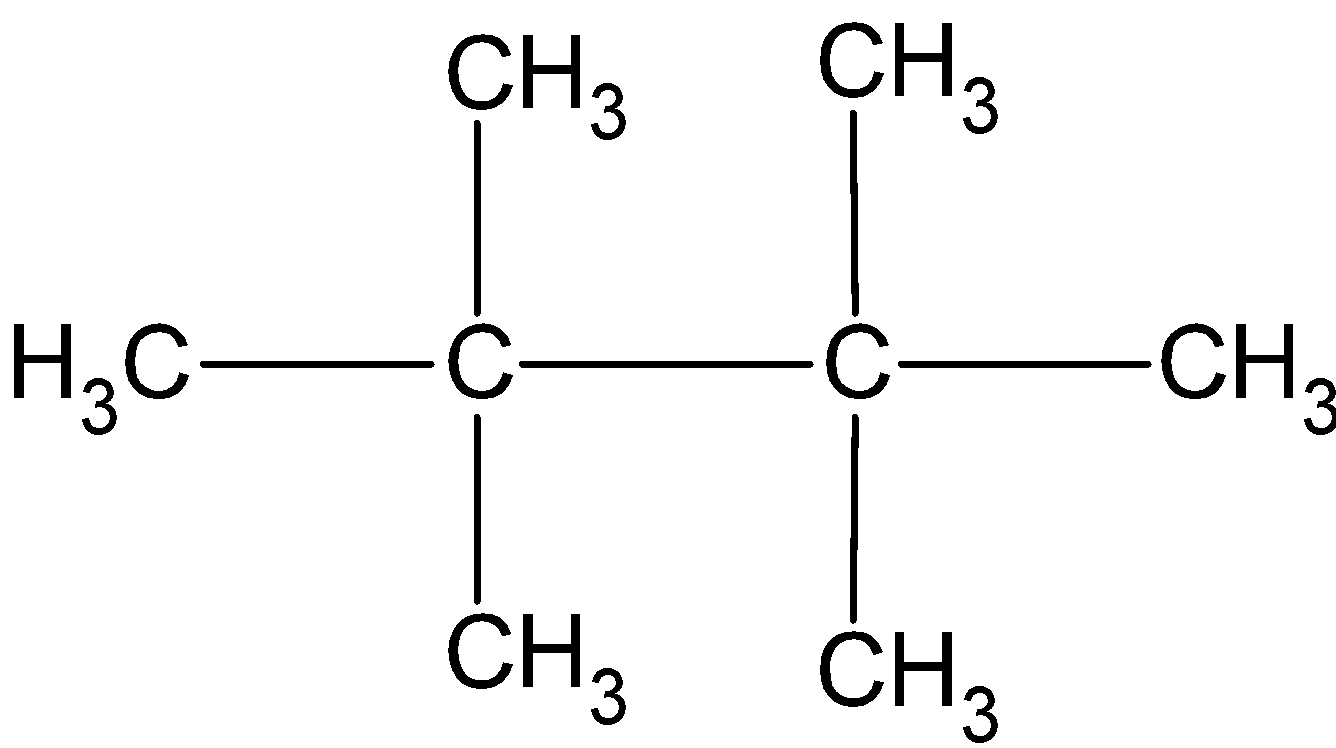
(B)
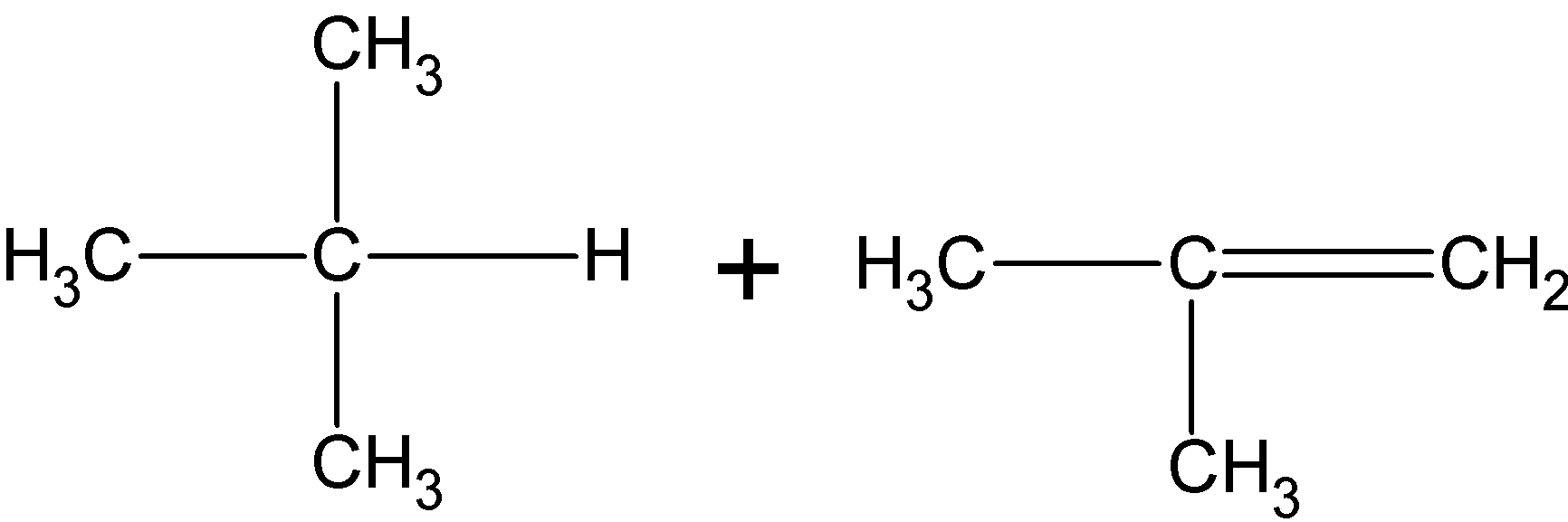
(C)
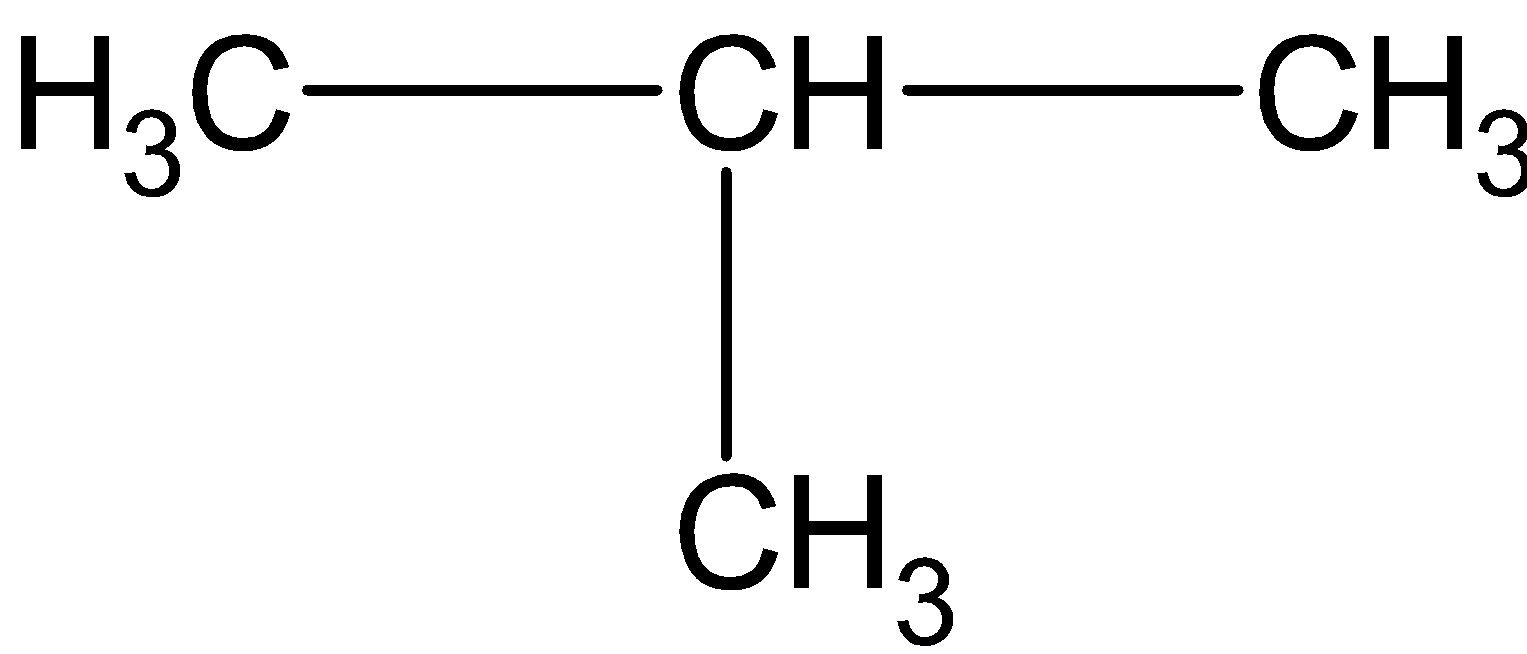
(D)




Answer
583.8k+ views
Hint: To answer this question, you must recall the reaction occurring when certain reagents are reacted with organic compounds. When an alkyl bromide is reacted with sodium in presence of ether, electrophilic substitution reaction occurs and this is a name reaction, known as the Williamson’s synthesis. You must also recall the tendencies of primary, secondary or tertiary halides to undergo substitution or electrophilic reactions.
Complete step by step solution:
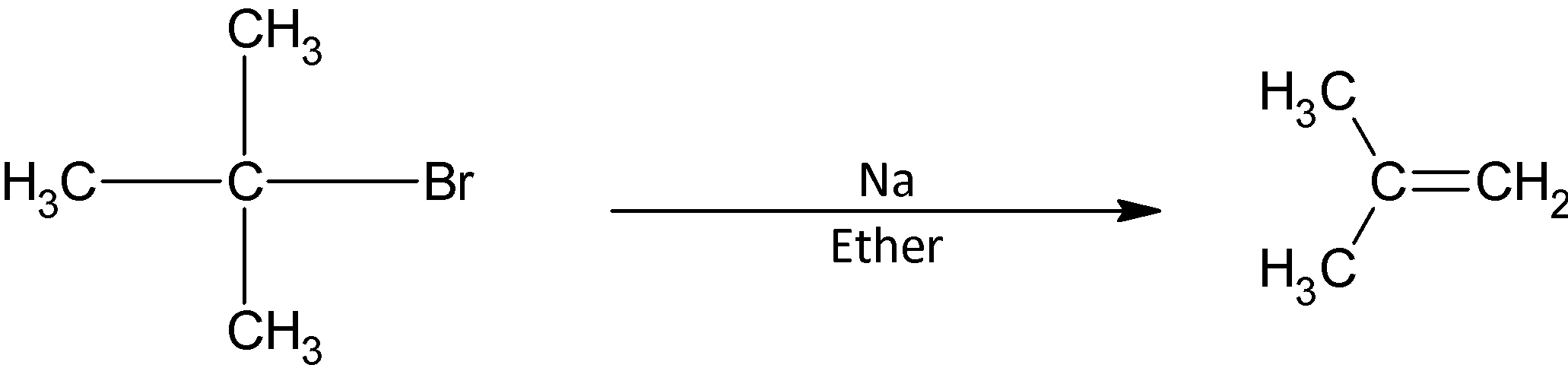
When alkyl halides undergo Williamson’s synthesis, bimolecular nucleophilic substitution reaction occurs and an ether is obtained as the product. The reacting species is sodium ethoxide. It is a one- step reaction and the addition of the nucleophile and removal of the bromide ion takes place in the same step.
However, in the given question, we have tertiary butoxide. We know that tertiary alkyl halides do not undergo bimolecular nucleophilic substitution reaction. There are three bulky methyl groups attached to the carbon atom susceptible to attack. The incoming ethoxide nucleophile is repelled by from the front side attack by the leaving bromide ion and its back side attack is hindered by the bulky methyl groups.
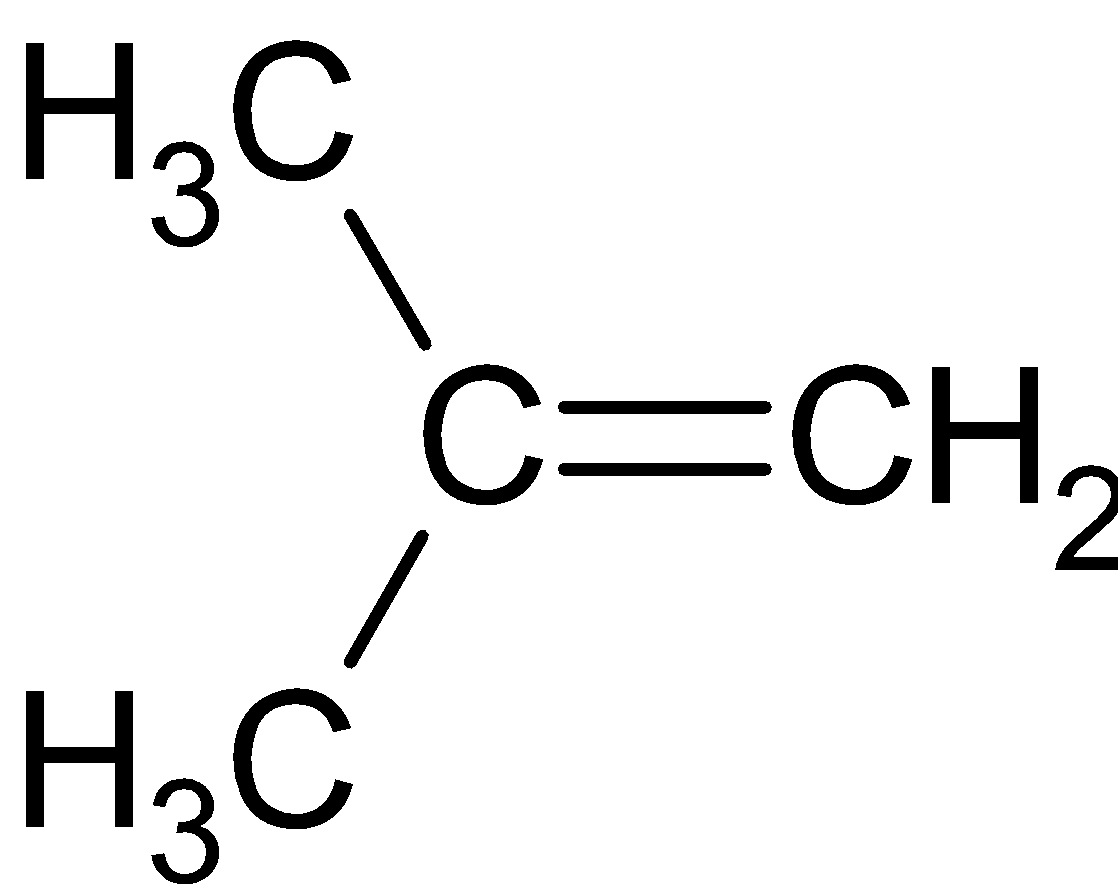
As a result, the ethoxide ion acts as a base in case of tertiary substrates. It removes a hydrogen ion from the adjacent methyl group. As a result, a double bond is formed between the two carbon atoms and the product obtained is an alkene. The product formed in case of tertiary butyl bromide.
The correct answer is (D).
Note:
Williamson's synthesis is a common reaction used for the preparation of ethers. Both symmetrical and unsymmetrical ethers can be prepared easily using this reaction.
Complete step by step solution:
When alkyl halides undergo Williamson’s synthesis, bimolecular nucleophilic substitution reaction occurs and an ether is obtained as the product. The reacting species is sodium ethoxide. It is a one- step reaction and the addition of the nucleophile and removal of the bromide ion takes place in the same step.
However, in the given question, we have tertiary butoxide. We know that tertiary alkyl halides do not undergo bimolecular nucleophilic substitution reaction. There are three bulky methyl groups attached to the carbon atom susceptible to attack. The incoming ethoxide nucleophile is repelled by from the front side attack by the leaving bromide ion and its back side attack is hindered by the bulky methyl groups.
As a result, the ethoxide ion acts as a base in case of tertiary substrates. It removes a hydrogen ion from the adjacent methyl group. As a result, a double bond is formed between the two carbon atoms and the product obtained is an alkene. The product formed in case of tertiary butyl bromide.

The correct answer is (D).
Note:
Williamson's synthesis is a common reaction used for the preparation of ethers. Both symmetrical and unsymmetrical ethers can be prepared easily using this reaction.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




