The number of $\sigma $ and $\pi $ -bonds in $F{{e}_{2}}{{(CO)}_{9}}$ , respectively are:
A. 22 $\sigma $ and 15 $\pi $
B. 23 $\sigma $ and 15 $\pi $
C. 22 $\sigma $ and 16 $\pi $
D. 15 $\sigma $ and 8 $\pi $
Answer
569.4k+ views
Hint: If p-orbitals are going to overlap axially then it leads to the formation of sigma ($\sigma $) bond and if p-orbitals are going to overlap sideways then it leads to the formation of the pi ($\pi $ ) bond. Sigma bond is stronger than pi bond.
Complete answer:
- In the question it is given to find the number of sigma bonds and number of pi bonds present in the given compound.
- The given compound is $F{{e}_{2}}{{(CO)}_{9}}$ .
- To know about how many sigma bonds and how many pi bonds present in the given compound we should know the structure of the given compound.
- The structure of the given compound is as follows.
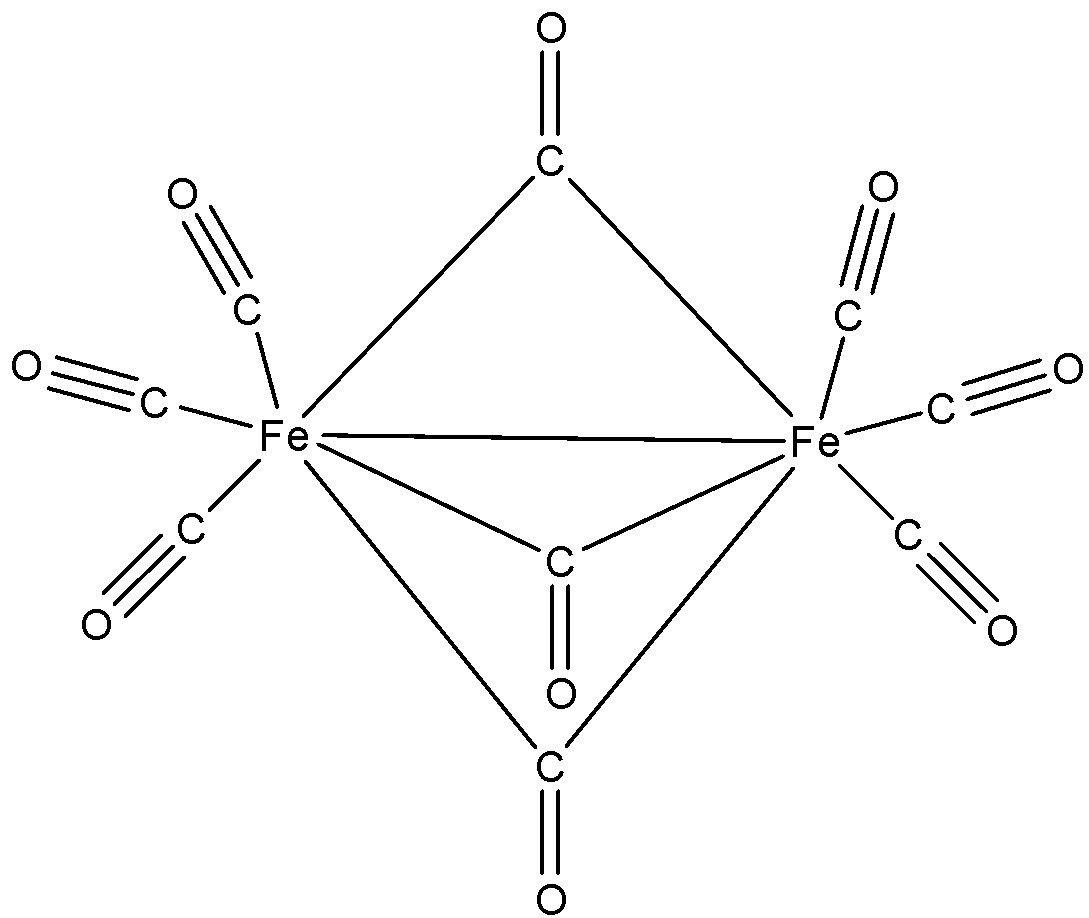
- All single bonds are sigma bonds only.
- In CO, there are three bonds. Out of those three one bond is sigma bond and the other two bonds are pi bonds.
- All Fe-CO bonds are sigma bonds in nature.
- If CO contains a double bond in the above structure then it contains one sigma bond and one pi bond in it.
- Therefore the number of sigma bonds present in the given compounds is 22 and the total number of pi bonds is 15.
So, the correct option is A.
Note:
Triple bonds are stronger than the double bond. Because in triple bonds there are two pi bonds and one pi bond while in a double bond there is one sigma bond and one pi bond. Therefore triple bonds are stronger than pi bonds.
Complete answer:
- In the question it is given to find the number of sigma bonds and number of pi bonds present in the given compound.
- The given compound is $F{{e}_{2}}{{(CO)}_{9}}$ .
- To know about how many sigma bonds and how many pi bonds present in the given compound we should know the structure of the given compound.
- The structure of the given compound is as follows.

- All single bonds are sigma bonds only.
- In CO, there are three bonds. Out of those three one bond is sigma bond and the other two bonds are pi bonds.
- All Fe-CO bonds are sigma bonds in nature.
- If CO contains a double bond in the above structure then it contains one sigma bond and one pi bond in it.
- Therefore the number of sigma bonds present in the given compounds is 22 and the total number of pi bonds is 15.
So, the correct option is A.
Note:
Triple bonds are stronger than the double bond. Because in triple bonds there are two pi bonds and one pi bond while in a double bond there is one sigma bond and one pi bond. Therefore triple bonds are stronger than pi bonds.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
How many of the following diseases can be studied with class 11 biology CBSE

Which of the following enzymes is used for carboxylation class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Receptor sites for neurotransmitters are present on class 11 biology CBSE

JGA JuxtaGlomerular Apparatus a sensitive region which class 11 biology CBSE

Root hairs develop from the region of A Meristematic class 11 biology CBSE




