The number of $P - O - P$ bridges in the structure of phosphorus pentoxide and phosphorus trioxide is respectively:
A.$6,6$
B.$5,5$
C.$5,6$
D.$6,5$
Answer
583.5k+ views
Hint: Phosphorus pentoxide and phosphorus trioxide are the two common oxides of phosphorus represented by the chemical formula $\left( {{P_4}{O_{10}}} \right)$ and $\left( {{P_4}{O_6}} \right)$ respectively. Phosphorus trioxide is represented as phosphorus hexoxide $\left( {{P_4}{O_6}} \right)$. The number of $P - O - P$ linkages or bridges can be easily calculated with the help of structure.
Complete step by step answer:
In the question, we are asked to calculate the number of $P - O - P$ bridges in the structure of phosphorus pentoxide and phosphorus trioxide. So, first, we will draw and analyze the structure of phosphorus pentoxide and phosphorus trioxide. So, now we will study the structure of the oxides one by one. Now let us consider the structure of phosphorus pentoxide $\left( {{P_4}{O_{10}}} \right)$. The structure is given below.
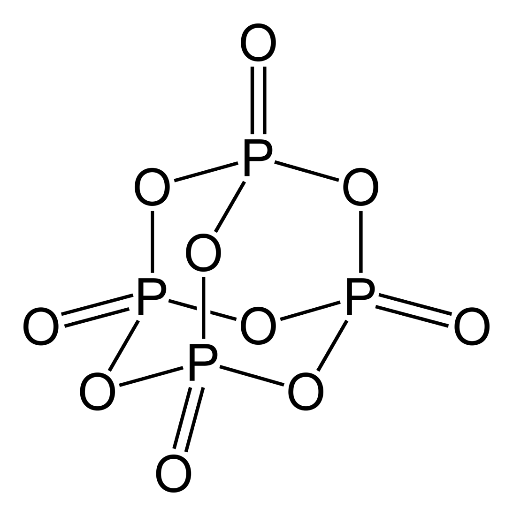
From the above structure of phosphorus pentoxide, we can observe that the total number of $P - O - P$ bridges in the structure of phosphorus pentoxide is $6$. Now we will consider the structure of phosphorus trioxide or hexoxide $\left( {{P_4}{O_6}} \right)$. The structure is given below.
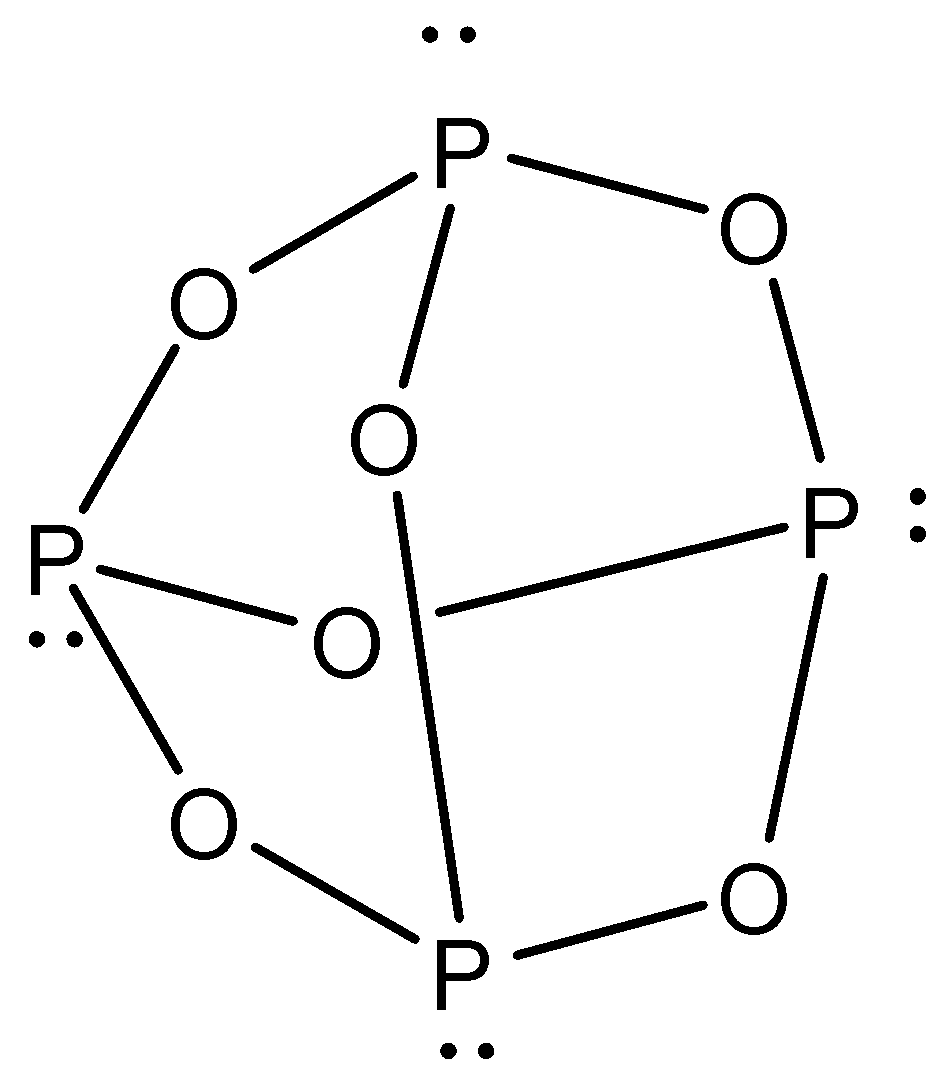
Again, from the above structure of phosphorus trioxide or hexoxide, we can observe that the total number of $P - O - P$ bridges in the structure of phosphorus trioxide or hexoxide is $6$. So, we can conclude that the total number of $P - O - P$ bridges in the structure of phosphorus pentoxide and phosphorus trioxide are $6,6$ respectively.
Therefore, the correct option is (A).
Note:
The phosphorus pentoxide is a white crystalline solid and it is the anhydride of phosphoric acid as well as acts as a powerful dehydrating agent. On the other hand, phosphorus trioxide is a colorless solid and it is the anhydride of phosphorous acid.
Complete step by step answer:
In the question, we are asked to calculate the number of $P - O - P$ bridges in the structure of phosphorus pentoxide and phosphorus trioxide. So, first, we will draw and analyze the structure of phosphorus pentoxide and phosphorus trioxide. So, now we will study the structure of the oxides one by one. Now let us consider the structure of phosphorus pentoxide $\left( {{P_4}{O_{10}}} \right)$. The structure is given below.

From the above structure of phosphorus pentoxide, we can observe that the total number of $P - O - P$ bridges in the structure of phosphorus pentoxide is $6$. Now we will consider the structure of phosphorus trioxide or hexoxide $\left( {{P_4}{O_6}} \right)$. The structure is given below.

Again, from the above structure of phosphorus trioxide or hexoxide, we can observe that the total number of $P - O - P$ bridges in the structure of phosphorus trioxide or hexoxide is $6$. So, we can conclude that the total number of $P - O - P$ bridges in the structure of phosphorus pentoxide and phosphorus trioxide are $6,6$ respectively.
Therefore, the correct option is (A).
Note:
The phosphorus pentoxide is a white crystalline solid and it is the anhydride of phosphoric acid as well as acts as a powerful dehydrating agent. On the other hand, phosphorus trioxide is a colorless solid and it is the anhydride of phosphorous acid.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




