The number of open chain isomers possible for ${C_4}{H_6}$:
A.$6$
B.$5$
C.$4$
D.$2$
Answer
555.5k+ views
Hint:In this question we will find the total number of open chain isomers possible for ${C_4}{H_6}$. We will use the basic concepts of isomerism and we will try to find open chain isomers only. We will exclude other isomers formed.
Complete step-by-step answer:First, we will understand the basic terms like ‘Isomers’ and ‘Open chain isomers’. So let’s understand these terms to find the exact number of open chain isomers possible for ${C_4}{H_6}$.
Isomers are molecules with the identical molecular formulas which means the same number of atoms of each element but the molecule will differ in the position of atoms and bonds. Now one more term ‘Open chain isomer’. It is one of the types of isomer in which a compound is acyclic with a linear structure rather than a cyclic one. An open chain isomer has no side chains.
Now we understand the basic terms used in the question. So we will find open chain isomers for ${C_4}{H_6}$.
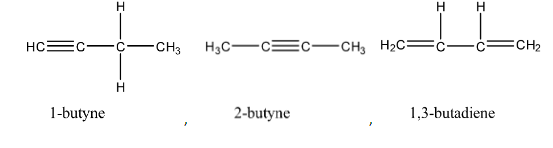
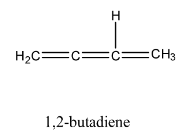
We can observe that the above molecules have identical molecular formula ${C_4}{H_6}$. Same number of carbon and hydrogen atoms but distinct arrangement of atoms and bonds.
In the question we are asked to find the total number of open chain isomers possible for ${C_4}{H_6}$. All above isomers are open chain because they are straight chain compounds, acyclic and isomers with no side chains.
We can observe that the total number of open chain isomers possible for ${C_4}{H_6}$ is $4$.
Therefore, the correct option is (C).
Note:The total number of isomers possible for ${C_4}{H_6}$ is $9$. Out of the $9$ isomers $4$ are open chain isomers and rest of them are cyclic isomers.
Isomers have identical molecular formula but their chemical and physical properties may not be the same.
Complete step-by-step answer:First, we will understand the basic terms like ‘Isomers’ and ‘Open chain isomers’. So let’s understand these terms to find the exact number of open chain isomers possible for ${C_4}{H_6}$.
Isomers are molecules with the identical molecular formulas which means the same number of atoms of each element but the molecule will differ in the position of atoms and bonds. Now one more term ‘Open chain isomer’. It is one of the types of isomer in which a compound is acyclic with a linear structure rather than a cyclic one. An open chain isomer has no side chains.
Now we understand the basic terms used in the question. So we will find open chain isomers for ${C_4}{H_6}$.


We can observe that the above molecules have identical molecular formula ${C_4}{H_6}$. Same number of carbon and hydrogen atoms but distinct arrangement of atoms and bonds.
In the question we are asked to find the total number of open chain isomers possible for ${C_4}{H_6}$. All above isomers are open chain because they are straight chain compounds, acyclic and isomers with no side chains.
We can observe that the total number of open chain isomers possible for ${C_4}{H_6}$ is $4$.
Therefore, the correct option is (C).
Note:The total number of isomers possible for ${C_4}{H_6}$ is $9$. Out of the $9$ isomers $4$ are open chain isomers and rest of them are cyclic isomers.
Isomers have identical molecular formula but their chemical and physical properties may not be the same.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Find the value of the expression given below sin 30circ class 11 maths CBSE

Name the Largest and the Smallest Cell in the Human Body ?

What organs are located on the left side of your body class 11 biology CBSE

What is the weight in lbs of a 75 Kg person class 11 physics CBSE

What is the percentage of photosynthetically active class 11 biology CBSE

How do I convert ms to kmh Give an example class 11 physics CBSE




