The molecule which does not exhibit dipole moment is:
(a)- $N{{H}_{3}}$
(b)- $CHC{{l}_{3}}$
(c)- ${{H}_{2}}O$
(d)- $CC{{l}_{4}}$
Answer
533.1k+ views
Hint: Draw the structure of all the compounds in the options and check their symmetry. To draw the dipole moment in the molecule, find the more electronegative atom joining the bond and draw the arrow towards the electronegative atom. If the molecule has a symmetry then the dipole will be zero.
Complete answer:
The dipole moment is calculated for the polar molecule, as the polar molecule has two different ends, i.e., a positive end and a negative end. The negative end represents the more electronegative atom and the positive end represents the less electronegative atom.
To draw the dipole moment in the molecule, find the more electronegative atom joining the bond and draw the arrow towards the electronegative atom. If the molecule has a symmetry then the dipole will be zero.
Let us draw all the structures of the compounds given in the question.
(a)- $N{{H}_{3}}$

This is not a symmetrical molecule and the dipole moment of $N{{H}_{3}}$ is non-zero. So, the dipole moment is 1.47D.
(b)- $CHC{{l}_{3}}$
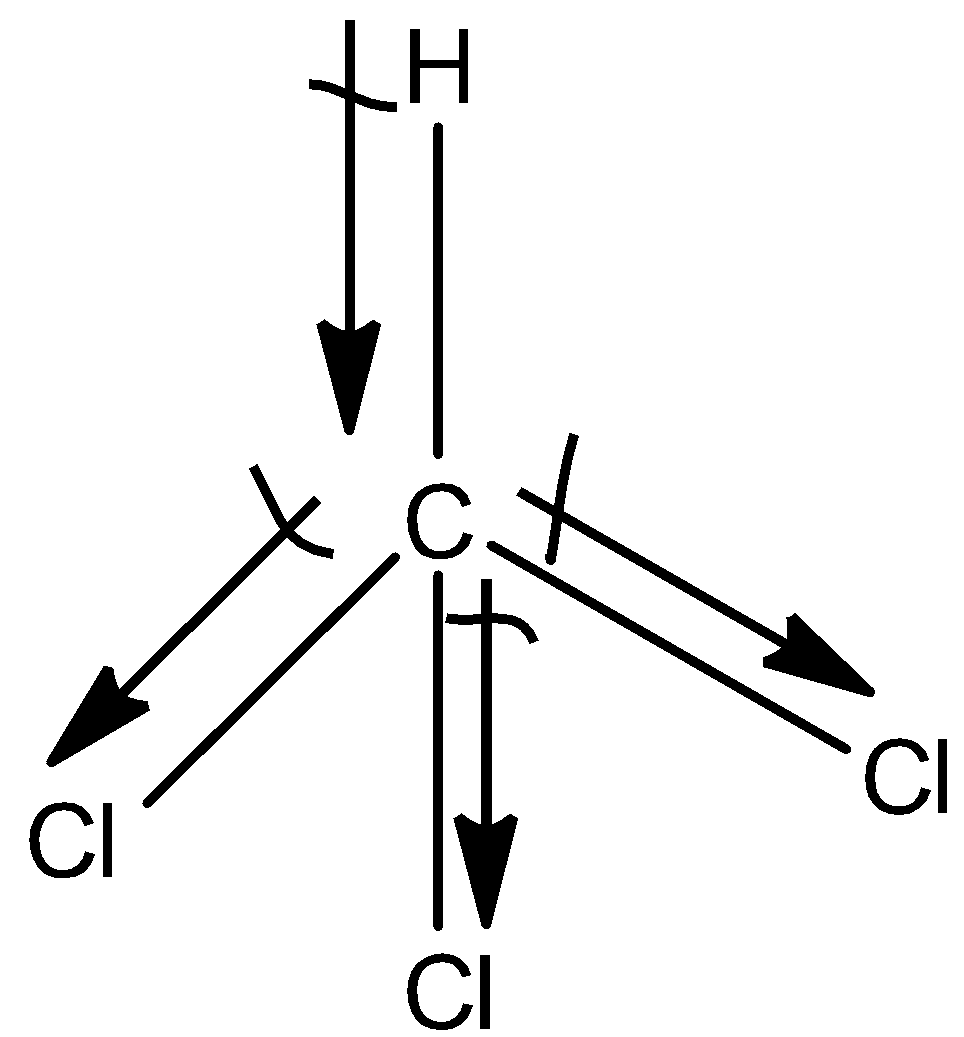
This is a symmetrical molecule but the atoms attached to the carbon atoms are different so, the dipole moment of $CHC{{l}_{3}}$ is also non-zero. So, the dipole moment is 1.04D.
(c)- ${{H}_{2}}O$
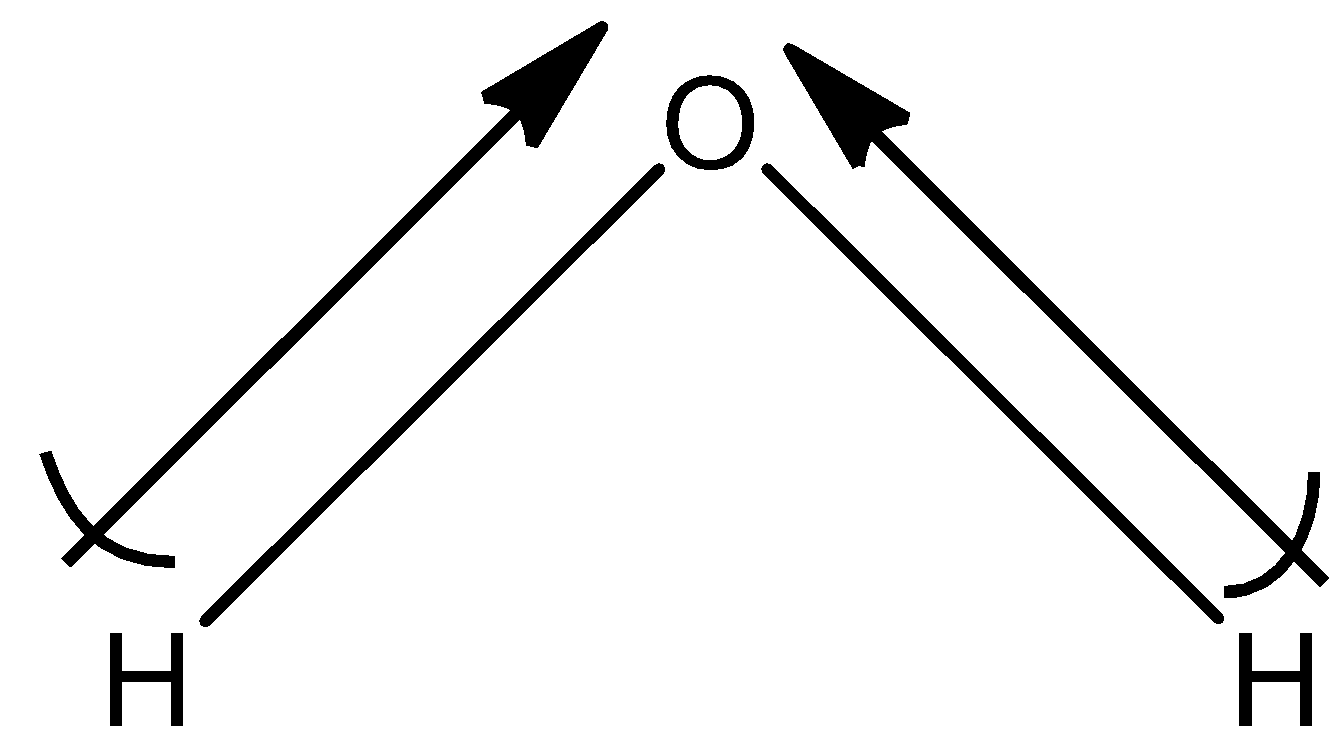
This is not a symmetrical molecule and the dipole moment of ${{H}_{2}}O$ is non-zero. So, the dipole moment is 1.84D.
(d)- $CC{{l}_{4}}$
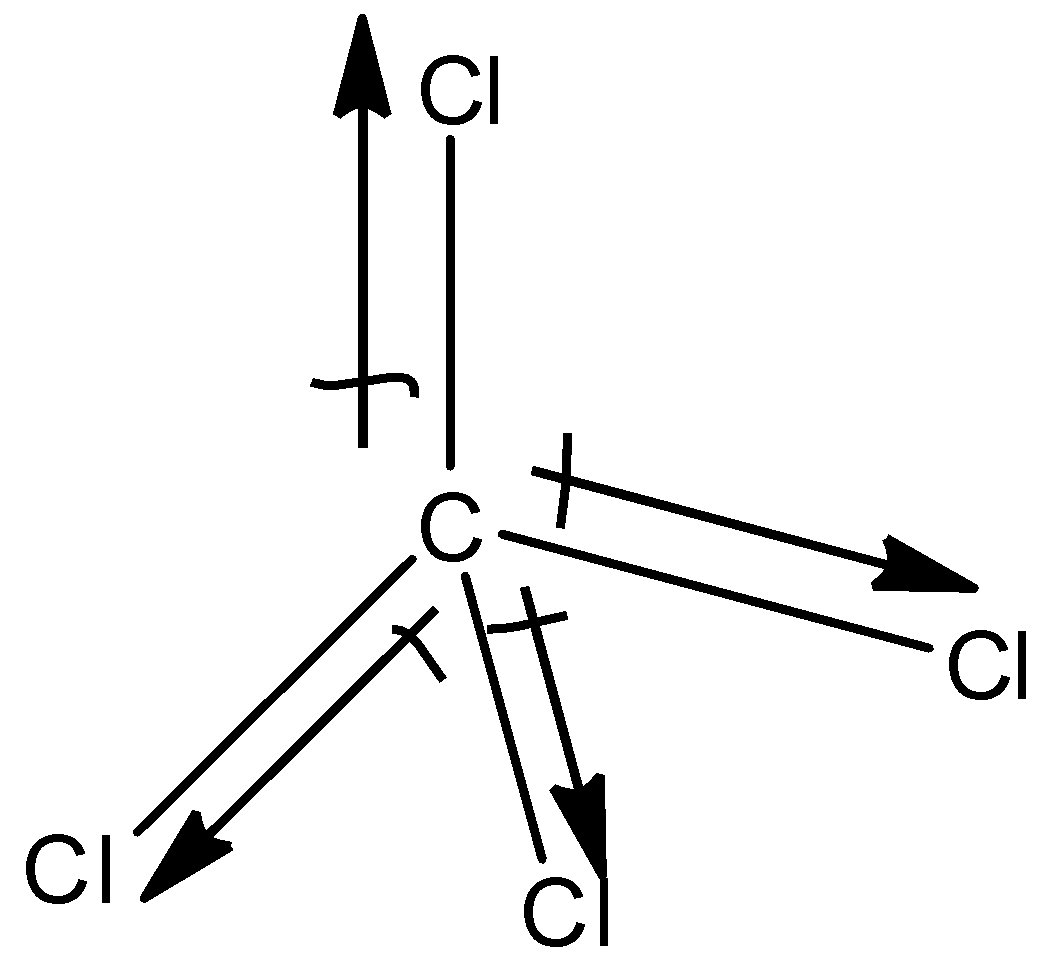
This is a symmetric molecule and all the atoms attached to the carbon atom are the same, so the dipole moment of $CC{{l}_{4}}$ is zero and it does not exhibit dipole moment.
Therefore, the correct answer is option (d)- $CC{{l}_{4}}$.
Note:
The dipole moment is defined mathematically as the product of the magnitude of a negative or positive charge (q) and the distance (d) between the centers of positive and negative charges mathematically.
Complete answer:
The dipole moment is calculated for the polar molecule, as the polar molecule has two different ends, i.e., a positive end and a negative end. The negative end represents the more electronegative atom and the positive end represents the less electronegative atom.
To draw the dipole moment in the molecule, find the more electronegative atom joining the bond and draw the arrow towards the electronegative atom. If the molecule has a symmetry then the dipole will be zero.
Let us draw all the structures of the compounds given in the question.
(a)- $N{{H}_{3}}$

This is not a symmetrical molecule and the dipole moment of $N{{H}_{3}}$ is non-zero. So, the dipole moment is 1.47D.
(b)- $CHC{{l}_{3}}$

This is a symmetrical molecule but the atoms attached to the carbon atoms are different so, the dipole moment of $CHC{{l}_{3}}$ is also non-zero. So, the dipole moment is 1.04D.
(c)- ${{H}_{2}}O$

This is not a symmetrical molecule and the dipole moment of ${{H}_{2}}O$ is non-zero. So, the dipole moment is 1.84D.
(d)- $CC{{l}_{4}}$

This is a symmetric molecule and all the atoms attached to the carbon atom are the same, so the dipole moment of $CC{{l}_{4}}$ is zero and it does not exhibit dipole moment.
Therefore, the correct answer is option (d)- $CC{{l}_{4}}$.
Note:
The dipole moment is defined mathematically as the product of the magnitude of a negative or positive charge (q) and the distance (d) between the centers of positive and negative charges mathematically.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

What organs are located on the left side of your body class 11 biology CBSE

Draw a labelled diagram showing structure of LS of class 11 biology CBSE

How many rings are present in MEDTA+n class 11 chemistry CBSE

Draw the molecular orbital diagram of N2N2 + N2 Write class 11 chemistry CBSE




