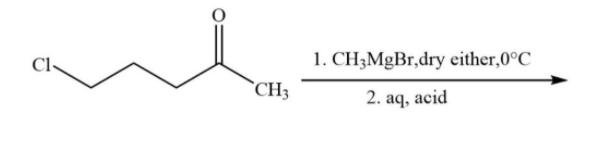
The major product in the following reaction is:
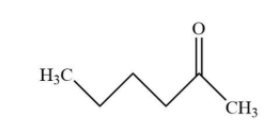
A.
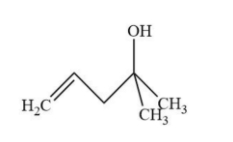
B.
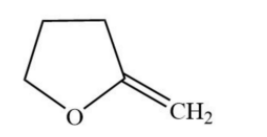
C.
D.
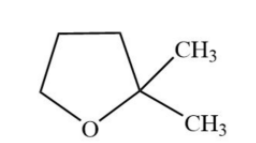





Answer
604.2k+ views
Hint: We know that $C{H_3}MgBr$ is known as Grignard reagent. The more electronegative an element is, the more it attracts the electron density in the bond. In the Grignard reagent, carbon of the alkyl group possesses negative charge and magnesium has positive charge.
Complete answer
Grignard reagents are made by reacting magnesium turnings with alkyl halides in ether solvents to form solutions of alkyl magnesium halide. Iodides, bromides, and chlorides can be used, as can both aryl and alkyl halides, though they cannot contain any functional groups that would react with the Grignard reagent once it is formed.
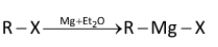
Where, R can be any alkyl/aryl while X is halogen.
Overall, it involves an insertion of magnesium into the new carbon–halogen bond. There is also a change in oxidation state of the magnesium, from Mg (0) to Mg (II). The reaction is therefore known as an oxidative insertion or oxidative addition.
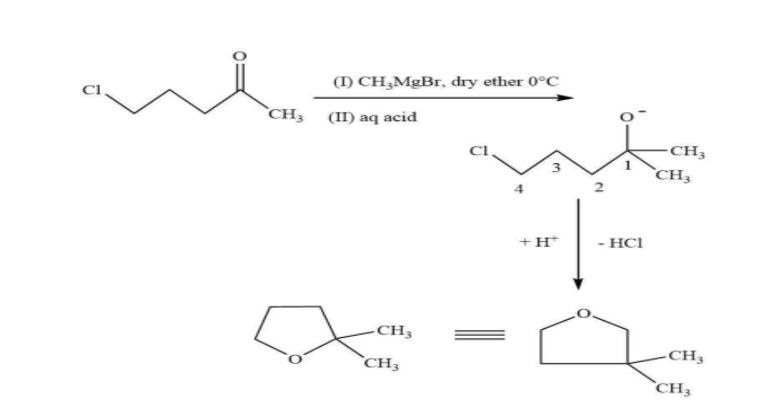
In this reaction, in the first step, the nucleophilic methyl group will attack on the electrophilic end of the carbonyl centre. Due to which a negative charge is generated on oxygen atom then in the second step there is a bimolecular substitution reaction on carbon containing chloride and chloride is removed. And overall, there is a formation of five membered compounds.
Hence, option (D) is the correct option.
Note:
Grignard reagents easily remove acidic protons. These strong nucleophilic reagents like Grignard reagents and organolithiums are strong bases, and may need protection from acidic protons as well as from electrophilic carbonyl groups. Among the most troublesome are the protons of hydroxyl groups.
Complete answer
Grignard reagents are made by reacting magnesium turnings with alkyl halides in ether solvents to form solutions of alkyl magnesium halide. Iodides, bromides, and chlorides can be used, as can both aryl and alkyl halides, though they cannot contain any functional groups that would react with the Grignard reagent once it is formed.

Where, R can be any alkyl/aryl while X is halogen.
Overall, it involves an insertion of magnesium into the new carbon–halogen bond. There is also a change in oxidation state of the magnesium, from Mg (0) to Mg (II). The reaction is therefore known as an oxidative insertion or oxidative addition.

In this reaction, in the first step, the nucleophilic methyl group will attack on the electrophilic end of the carbonyl centre. Due to which a negative charge is generated on oxygen atom then in the second step there is a bimolecular substitution reaction on carbon containing chloride and chloride is removed. And overall, there is a formation of five membered compounds.
Hence, option (D) is the correct option.
Note:
Grignard reagents easily remove acidic protons. These strong nucleophilic reagents like Grignard reagents and organolithiums are strong bases, and may need protection from acidic protons as well as from electrophilic carbonyl groups. Among the most troublesome are the protons of hydroxyl groups.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




