The IUPAC nomenclature of the given compound is:
$C{H_2} = C(C{H_3}) - C{H_2} - C{(C{H_3})_2} - CO - C{H_2} - C{H_3}$
Answer
600k+ views
Hint:The given compound contains an alkene group as well as a ketone group. According to the IUPAC priority table, the preference of the ketone group is more than that of the preference of alkene functional group and when we name a compound which is containing a ketone functional group then the ‘one’ is used as a suffix.
Complete step by step answer:
As we can see in the given compound there are two functional groups present that are alkene functional groups that is $C = C$ and also a ketone functional group that is $R - CO - R'$. In such conditions when we have more than one functional group in a compound then the compound which has higher priority according to IUPAC priority order will be named first and other groups will be considered as substituents. According to IUPAC, priority order can be given as:
Aldehyde$ > $ketone$ > $alcohol$ > $alkene
As we can see that the ketone functional group has higher priority than the ethene functional group therefore, we will start numbering so that the ketone group gets the lowest number. The numbering of the compound can be given as:
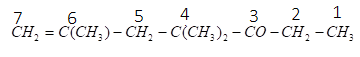
For ketone functional groups, we use ‘one’ as the suffix and for the ethene group we use ‘ene’ as a suffix. As the longest carbon chain includes seven carbons and at carbon number $4$ , there are two methyl groups (we will use dimethyl) and at carbon number $6$, there is one methyl group (we will use only methyl) and one ethene group. So, we will name substituent in alphabetical order. Therefore, the name of the given compound can be written as:
$4 - $dimethyl $ - 6 - $methyl$ - 6 - $hepten$ - 3 - $one.
Note:
Always remember that if in a compound there are more than one functional group in that compound then the functional group with the higher priority is named first and the other groups are taken as a substituent.
Complete step by step answer:
As we can see in the given compound there are two functional groups present that are alkene functional groups that is $C = C$ and also a ketone functional group that is $R - CO - R'$. In such conditions when we have more than one functional group in a compound then the compound which has higher priority according to IUPAC priority order will be named first and other groups will be considered as substituents. According to IUPAC, priority order can be given as:
Aldehyde$ > $ketone$ > $alcohol$ > $alkene
As we can see that the ketone functional group has higher priority than the ethene functional group therefore, we will start numbering so that the ketone group gets the lowest number. The numbering of the compound can be given as:
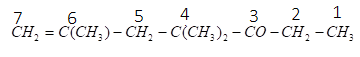
For ketone functional groups, we use ‘one’ as the suffix and for the ethene group we use ‘ene’ as a suffix. As the longest carbon chain includes seven carbons and at carbon number $4$ , there are two methyl groups (we will use dimethyl) and at carbon number $6$, there is one methyl group (we will use only methyl) and one ethene group. So, we will name substituent in alphabetical order. Therefore, the name of the given compound can be written as:
$4 - $dimethyl $ - 6 - $methyl$ - 6 - $hepten$ - 3 - $one.
Note:
Always remember that if in a compound there are more than one functional group in that compound then the functional group with the higher priority is named first and the other groups are taken as a substituent.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which gas is abundant in air class 11 chemistry CBSE




