
The IUPAC name of the compound is:
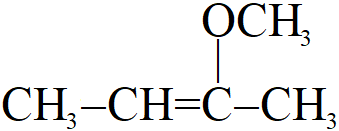
A. 1-Methoxy-1-methyl propene
B. 2-methoxy but-2-ene
C. Dimethyl propene ether
D. None of these
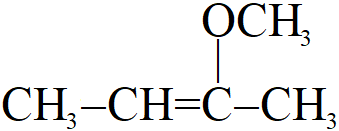
Answer
604.5k+ views
Hint: In order to determine the IUPAC name of given carbon structure first we will understand what is the process to give an IUPAC name of any carbon structure then according to it we will write the IUPAC name of given structure.
Complete step-by-step answer:
Here is a short set of guidelines for writing the name of every carbon structure in IUPAC.
The longest carbon chain is known as the parent line.
Identify all substituent classes (attached from the parent chain).
Number the parent chain carbons from the end and give the lowest number to the substituent. The series which is the "lowest" when evaluating a set of numbers is the one where the first difference contains the lowest number. If two or more lateral chains are equal, assign the lowest number to the first one in the description.
If the same substituent appears more than once, the position for every point where the substituent appears is indicated. Alternatively, the number of times the substituent category occurs is defined by a suffix (di, tri, tetra etc.)
If two or more different substituents exist, they are identified in alphabetical order using the base name (the prefixes are neglected).
Apart from these rules there are some more rules as well.
Here from the structure we find the number of carbon in the longest chain is 4 so the name will be contain “but” and also the chain with 4 carbon has a double bond so, its name on the basis of longest chain will be “butene” as the double bond is at second carbon so the name “but-2-ene”.
The compound has a “methoxy” group attached to the second carbon from the right. So the prefix will be “2-methoxy”
Hence, the given compound is “2-methoxy but-2-ene”.
So, option B is the correct option.
Note- The IUPAC nomenclature in organic chemistry is a system in organic chemical compounds in molecular nomenclature, as defined by the International Union of Pure and Applied Chemistry (IUPAC). This is written in the Organic Chemistry Nomenclature (informally called the Blue Book). Ideally a conceivable organic compound should have a name from which to construct an unmistakable structural formula. Some of the rules are listed in the solution, to name a compound, students must remember certain rules.
Complete step-by-step answer:
Here is a short set of guidelines for writing the name of every carbon structure in IUPAC.
The longest carbon chain is known as the parent line.
Identify all substituent classes (attached from the parent chain).
Number the parent chain carbons from the end and give the lowest number to the substituent. The series which is the "lowest" when evaluating a set of numbers is the one where the first difference contains the lowest number. If two or more lateral chains are equal, assign the lowest number to the first one in the description.
If the same substituent appears more than once, the position for every point where the substituent appears is indicated. Alternatively, the number of times the substituent category occurs is defined by a suffix (di, tri, tetra etc.)
If two or more different substituents exist, they are identified in alphabetical order using the base name (the prefixes are neglected).
Apart from these rules there are some more rules as well.
Here from the structure we find the number of carbon in the longest chain is 4 so the name will be contain “but” and also the chain with 4 carbon has a double bond so, its name on the basis of longest chain will be “butene” as the double bond is at second carbon so the name “but-2-ene”.
The compound has a “methoxy” group attached to the second carbon from the right. So the prefix will be “2-methoxy”
Hence, the given compound is “2-methoxy but-2-ene”.
So, option B is the correct option.
Note- The IUPAC nomenclature in organic chemistry is a system in organic chemical compounds in molecular nomenclature, as defined by the International Union of Pure and Applied Chemistry (IUPAC). This is written in the Organic Chemistry Nomenclature (informally called the Blue Book). Ideally a conceivable organic compound should have a name from which to construct an unmistakable structural formula. Some of the rules are listed in the solution, to name a compound, students must remember certain rules.
Recently Updated Pages
Master Class 10 Computer Science: Engaging Questions & Answers for Success

Master Class 10 General Knowledge: Engaging Questions & Answers for Success

Master Class 10 English: Engaging Questions & Answers for Success

Master Class 10 Social Science: Engaging Questions & Answers for Success

Master Class 10 Maths: Engaging Questions & Answers for Success

Master Class 10 Science: Engaging Questions & Answers for Success

Trending doubts
What is the median of the first 10 natural numbers class 10 maths CBSE

Which women's tennis player has 24 Grand Slam singles titles?

Who is the Brand Ambassador of Incredible India?

Why is there a time difference of about 5 hours between class 10 social science CBSE

Write a letter to the principal requesting him to grant class 10 english CBSE

A moving boat is observed from the top of a 150 m high class 10 maths CBSE




