The gas liberated by heating potassium permanganate (\[KMn{{O}_{4}}\]) is?
(A) Hydrogen
(B) Oxygen
(C) Nitrogen
(D) Methane
Answer
612.3k+ views
Hint: Whenever we are heating any chemical then the chemical converts from one form to other forms by liberating some gas. Potassium permanganate (\[KMn{{O}_{4}}\]) is a chemical that exists in the solid-state at room temperature.
Complete step by step solution:
-The structure of the potassium permanganate is as follows.
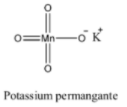
-The reaction of heating of potassium permanganate is as follows.
\[KMn{{O}_{4}}\xrightarrow{\Delta }{{K}_{2}}Mn{{O}_{4}}+Mn{{O}_{2}}+{{O}_{2}}\]
-On heating, potassium permanganate gives potassium manganate, Manganese dioxide and oxygen as the products.
-Coming to given options, option A Hydrogen. In the molecular formula of potassium permanganate (\[KMn{{O}_{4}}\]), there is no hydrogen. So, on heating potassium permanganate, hydrogen gas is not going to form as one of the product. Hence option A is wrong.
-Coming option B, Oxygen. In the molecular formula of potassium permanganate (\[KMn{{O}_{4}}\]), there is a presence of oxygen. So, on heating potassium permanganate, oxygen gas is going to form as one of the product. Hence option B is correct.
-Coming to option C, Nitrogen. In the molecular formula of potassium permanganate (\[KMn{{O}_{4}}\]), there is no nitrogen. So, on heating potassium permanganate, Nitrogen gas is not going to form as one of the product. Hence option C is wrong.
-Coming to option D, Methane. In the molecular formula of potassium permanganate (\[KMn{{O}_{4}}\]) there is no Carbon and hydrogen. So, on heating potassium permanganate, Methane gas is not going to form as one of the product. Hence option D is wrong.
-Therefore on heating potassium permanganate, oxygen gas is going to form as one of the product.
So, the correct option is (B).
Note: The oxidation state of Manganese in potassium permanganate (\[KMn{{O}_{4}}\]) is +7. On heating potassium permanganate (\[KMn{{O}_{4}}\]) gives potassium manganate (\[{{K}_{2}}Mn{{O}_{4}}\]). The oxidation state of manganese in \[{{K}_{2}}Mn{{O}_{4}}\] is +6.
Complete step by step solution:
-The structure of the potassium permanganate is as follows.

-The reaction of heating of potassium permanganate is as follows.
\[KMn{{O}_{4}}\xrightarrow{\Delta }{{K}_{2}}Mn{{O}_{4}}+Mn{{O}_{2}}+{{O}_{2}}\]
-On heating, potassium permanganate gives potassium manganate, Manganese dioxide and oxygen as the products.
-Coming to given options, option A Hydrogen. In the molecular formula of potassium permanganate (\[KMn{{O}_{4}}\]), there is no hydrogen. So, on heating potassium permanganate, hydrogen gas is not going to form as one of the product. Hence option A is wrong.
-Coming option B, Oxygen. In the molecular formula of potassium permanganate (\[KMn{{O}_{4}}\]), there is a presence of oxygen. So, on heating potassium permanganate, oxygen gas is going to form as one of the product. Hence option B is correct.
-Coming to option C, Nitrogen. In the molecular formula of potassium permanganate (\[KMn{{O}_{4}}\]), there is no nitrogen. So, on heating potassium permanganate, Nitrogen gas is not going to form as one of the product. Hence option C is wrong.
-Coming to option D, Methane. In the molecular formula of potassium permanganate (\[KMn{{O}_{4}}\]) there is no Carbon and hydrogen. So, on heating potassium permanganate, Methane gas is not going to form as one of the product. Hence option D is wrong.
-Therefore on heating potassium permanganate, oxygen gas is going to form as one of the product.
So, the correct option is (B).
Note: The oxidation state of Manganese in potassium permanganate (\[KMn{{O}_{4}}\]) is +7. On heating potassium permanganate (\[KMn{{O}_{4}}\]) gives potassium manganate (\[{{K}_{2}}Mn{{O}_{4}}\]). The oxidation state of manganese in \[{{K}_{2}}Mn{{O}_{4}}\] is +6.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Explain the Treaty of Vienna of 1815 class 10 social science CBSE

Which Indian city is known as the "City of Victory"?

Which instrument is used to measure the Blood Pressure?

Who Won 36 Oscar Awards? Record Holder Revealed

What is the median of the first 10 natural numbers class 10 maths CBSE

Why is it 530 pm in india when it is 1200 afternoon class 10 social science CBSE




