
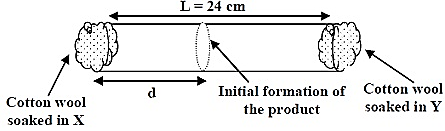
The experimental value of d is found to be smaller than the estimate obtained using Graham's law. This is due to the:
A. Larger mean free path for X as compared to that of Y
B. Larger mean free path for Y as compared to that of X
C. increased collision frequency of Y with the inert gas as compared to that X with the inert gas.
D. increased collision frequency of X with the inert gas as compared to that of Y with inert.

Answer
588.9k+ views
Hint: For this problem, we have to study Graham's law of diffusion and the relation between the mean free path and collision frequency. After which we can determine the reason behind the small value of d.
Complete step by step answer:
- In the given question, we have to explain the reason behind the smaller value of d by using Graham's law of diffusion.
- Now, we know that according to Graham's law of diffusion, the rate of effusion of the gases of the two gases has the inverse effect on the square root of the molecular mass of the gas.
- Also, we know that collision frequency is defined as the average rate of the collision of between two reactant molecules per unit time in a given volume of the container.
- So, when the molecules are present close to each other, the rate of the collision frequency increases.
- The increase in the collision frequency of the X molecules occurs with the inert gas molecules due to which the mean free path decreases.
- Now, due to the decreases in the mean free path of the molecules, the molecular speed of the gaseous molecules also decreases.
- The molecular decreases because the distance required for the collision also decreases.
- Thus, the value of d is smaller as compared to that of the Y.
So, the correct answer is “Option D”.
Note: In the problem, the mean free path of the gas particles is defined as the average distance travelled that can be travelled by the colliding particles. The temperature affects the mean free path because when the temperature increases the kinetic energy of the molecules also increases due to which the collision frequency will also increase.
Complete step by step answer:
- In the given question, we have to explain the reason behind the smaller value of d by using Graham's law of diffusion.
- Now, we know that according to Graham's law of diffusion, the rate of effusion of the gases of the two gases has the inverse effect on the square root of the molecular mass of the gas.
- Also, we know that collision frequency is defined as the average rate of the collision of between two reactant molecules per unit time in a given volume of the container.
- So, when the molecules are present close to each other, the rate of the collision frequency increases.
- The increase in the collision frequency of the X molecules occurs with the inert gas molecules due to which the mean free path decreases.
- Now, due to the decreases in the mean free path of the molecules, the molecular speed of the gaseous molecules also decreases.
- The molecular decreases because the distance required for the collision also decreases.
- Thus, the value of d is smaller as compared to that of the Y.
So, the correct answer is “Option D”.
Note: In the problem, the mean free path of the gas particles is defined as the average distance travelled that can be travelled by the colliding particles. The temperature affects the mean free path because when the temperature increases the kinetic energy of the molecules also increases due to which the collision frequency will also increase.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Explain zero factorial class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

What steps did the French revolutionaries take to create class 11 social science CBSE

The transition element that has lowest enthalpy of class 11 chemistry CBSE

Can anyone list 10 advantages and disadvantages of friction




