
The electronic configuration which obeys Hund’s rule for the ground state of a carbon atom?
A.
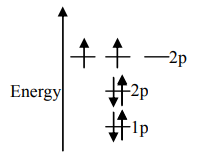
B.
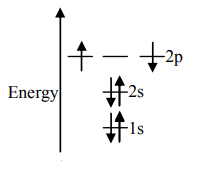
C.
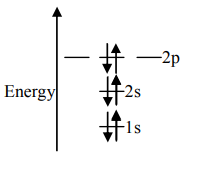
D.
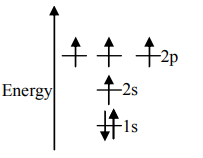




Answer
514.8k+ views
Hint :As per Hund’s rule all every orbital is singly filled before double occupancy takes place. All electrons in the single occupancy have the same spin; this gives maximum total spin. In half-filled orbits, electrons will not pair with any other electron as it has to fill all the orbits singly first.
Complete Step By Step Answer:
Unpaired electrons present in the orbit are at the ground state. As per Hund’s rule of maximum multiplicity, the term with maximum multiplicity falls lowest in the energy. Now as per the rule all the orbits should be first singly filled in option A the configuration is $1{s^2}2{s^2}2{p^2}$, here as we can see the $1s$orbit is completely filled with opposite charges, also the $2s$ orbit is completely filled with opposite charge but the $2p$ orbital is filled with an only single charge as it has to singly fill all the orbits first. In $2p$ the orbit is filled with the same charge that is either $ - $or $ + $. This completely follows Hund’s rule. While in option B first and the last orbit is singly filled keeping the center orbit empty violating Hund’s rule of maximum multiplicity. Similarly in option C the center orbit is completely filled keeping the other orbits empty.
Therefore the correct option is A.
Note :
Hund’s rule is mainly used in chemistry, quantum physics, and spectroscopy. As per Hund’s rule of maximum multiplicity pairing in p d, f orbitals cannot occur until all the orbits are first singly filled. Electrons repel each other as they are negatively charged.
Complete Step By Step Answer:
Unpaired electrons present in the orbit are at the ground state. As per Hund’s rule of maximum multiplicity, the term with maximum multiplicity falls lowest in the energy. Now as per the rule all the orbits should be first singly filled in option A the configuration is $1{s^2}2{s^2}2{p^2}$, here as we can see the $1s$orbit is completely filled with opposite charges, also the $2s$ orbit is completely filled with opposite charge but the $2p$ orbital is filled with an only single charge as it has to singly fill all the orbits first. In $2p$ the orbit is filled with the same charge that is either $ - $or $ + $. This completely follows Hund’s rule. While in option B first and the last orbit is singly filled keeping the center orbit empty violating Hund’s rule of maximum multiplicity. Similarly in option C the center orbit is completely filled keeping the other orbits empty.
Therefore the correct option is A.
Note :
Hund’s rule is mainly used in chemistry, quantum physics, and spectroscopy. As per Hund’s rule of maximum multiplicity pairing in p d, f orbitals cannot occur until all the orbits are first singly filled. Electrons repel each other as they are negatively charged.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

The largest wind power cluster is located in the state class 11 social science CBSE

Explain zero factorial class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Which among the following are examples of coming together class 11 social science CBSE

Can anyone list 10 advantages and disadvantages of friction




