The correct name for is:
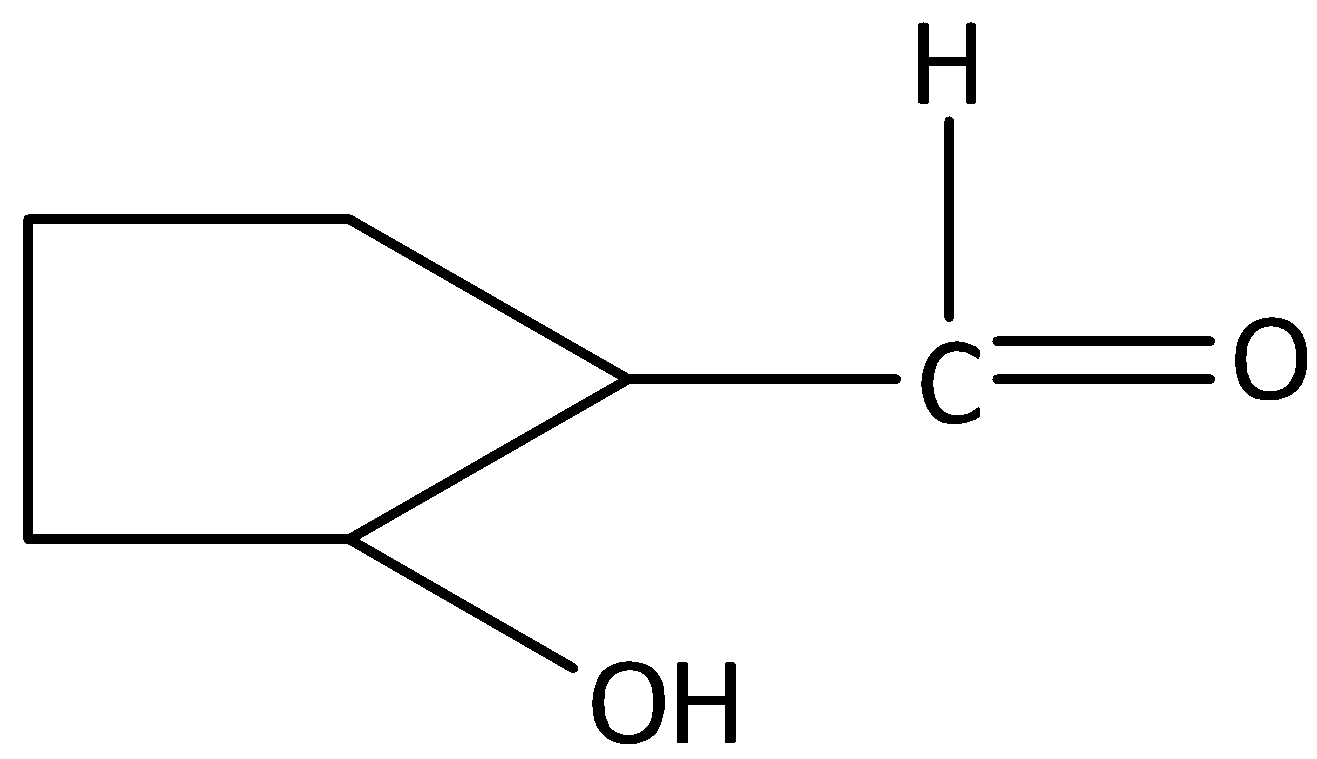
A. 2-Hydroxycyclopentane-1-al
B. 2-Formula-1-hydroxycyclopentane
C. 2-Hydroxycyclopentane Carbon Aldehyde
D. Cyclopentane-2-ol-1-al

Answer
611.7k+ views
Hint: Aldehydes are organic compounds that contain carbonyl groups. We can write the general formula of aldehydes as\[R - CHO\], here R is the alkyl group. Aldehydes are used in the preparation of plasticizers and polyols, they can be reduced to alcohols.
Complete step by step answer: Aldehydes contain the carbonyl group.
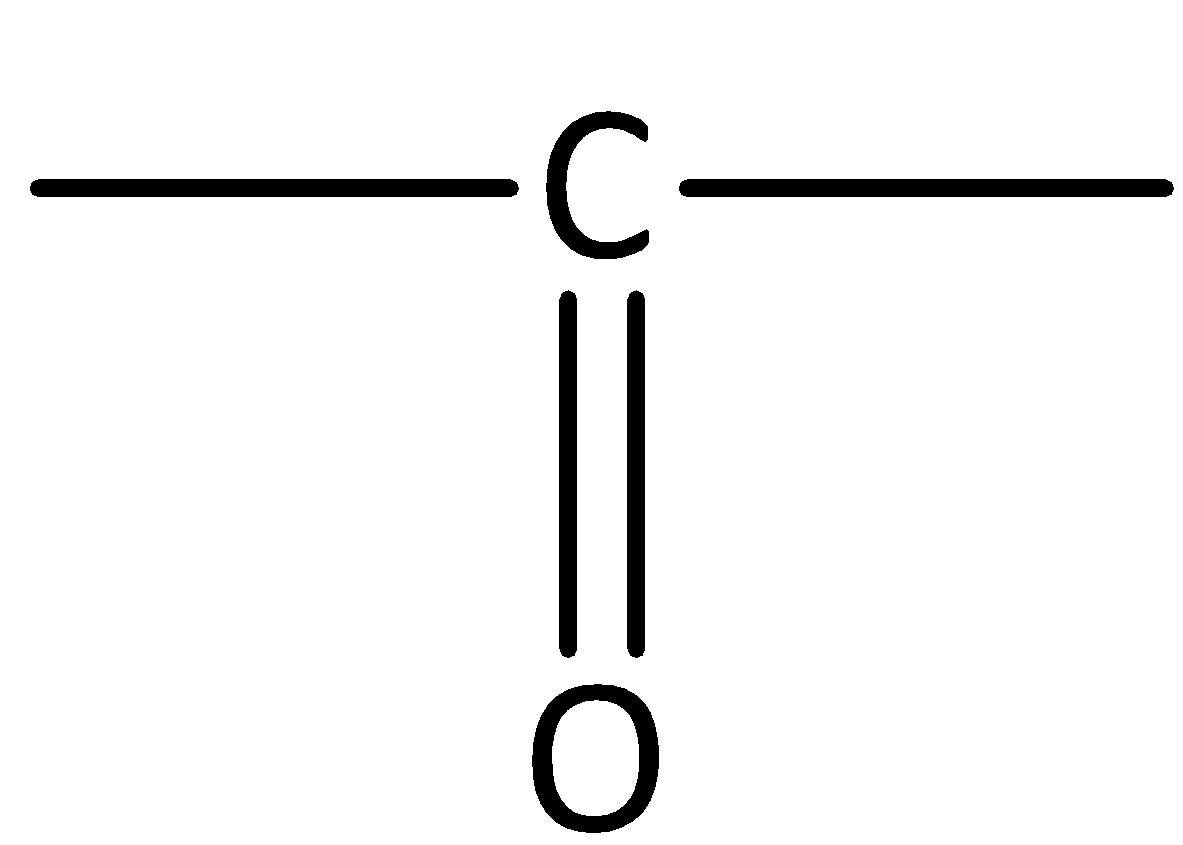
We can give the IUPAC Nomenclature for naming aldehydes as follows,
The parent compound; that is the longest continuous carbon chain containing the carbonyl group has to be determined.
Replace the final –e of the parent alkane with –al.
The chain beginning with the carbonyl carbon is named as carbon${\text{ - 1}}{\text{.}}$
All the substituents are named and numbered.
No number is used for the position of the carbonyl group because it is always at the end of the parent chain. Therefore, it must be carbon${\text{ - 1}}{\text{.}}$
We can give the common name system for naming aldehydes as follows,
- The common names of the aldehydes are derived from the Latin roots.
- Example, the common name of methanal is formaldehyde.
- Common name of substituted aldehydes can be given as follows,
- The common names of substituted aldehydes are named as derivatives of straight-chain parent compounds.
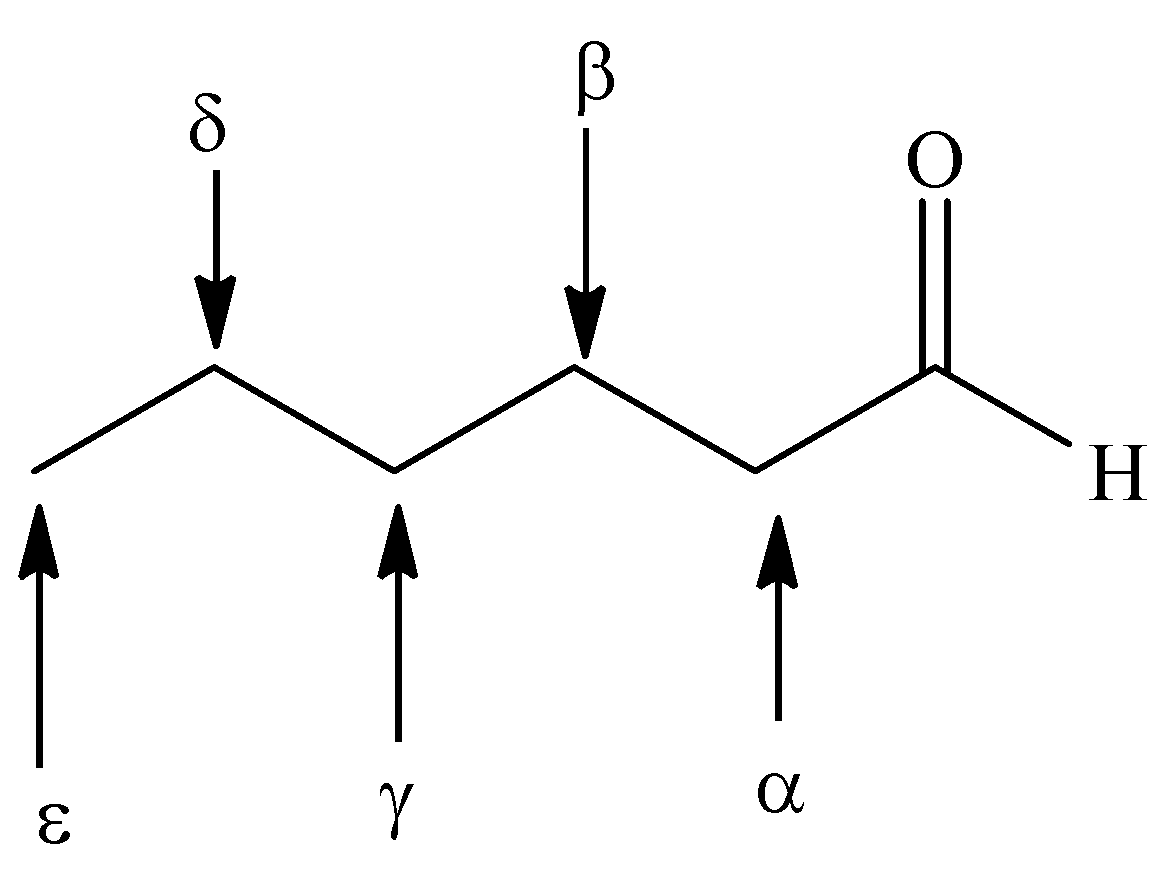
- Greek letters designate the carbon atoms near the carbonyl group.
- The carbon atom bonded to the carbonyl group is alpha, the next removed is beta and so on.
We use the suffix -carbaldehyde, when the carbonyl group \[\left( { - CHO} \right)\]is linked to the ring.
We can draw the structure of the given aldehyde as,
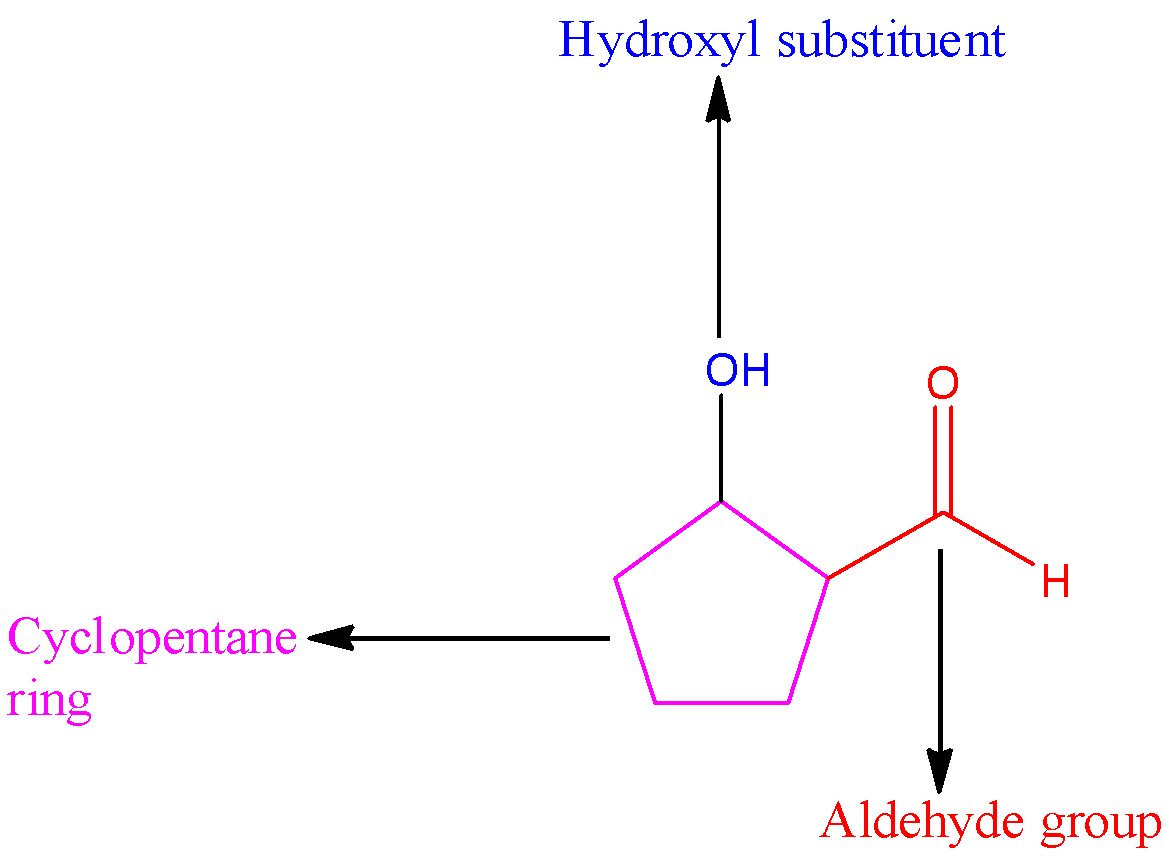
The parent carbon chain in the given compound is cyclopentane, the substituent is a hydroxyl group present at carbon in the second position. The carbonyl carbon will occupy the first position. Since the aldehyde group is attached to the cyclopentane ring, the name of the compound is 2-hydroxycyclopentane carbaldehyde.
So, the correct answer is “Option C”.
Note: We can prepare carboxylic acids by oxidizing aldehydes. We can also prepare alcohol by reducing aldehydes. Smaller aldehydes are sparingly water soluble, higher aldehydes are insoluble in water. Aldehydes are used in organic reactions such as,
- Woff-Kishner reduction
- Pinacol coupling reaction
- Oxo-Diels-Alder reaction
- Decarbonylation
Complete step by step answer: Aldehydes contain the carbonyl group.

We can give the IUPAC Nomenclature for naming aldehydes as follows,
The parent compound; that is the longest continuous carbon chain containing the carbonyl group has to be determined.
Replace the final –e of the parent alkane with –al.
The chain beginning with the carbonyl carbon is named as carbon${\text{ - 1}}{\text{.}}$
All the substituents are named and numbered.
No number is used for the position of the carbonyl group because it is always at the end of the parent chain. Therefore, it must be carbon${\text{ - 1}}{\text{.}}$
We can give the common name system for naming aldehydes as follows,
- The common names of the aldehydes are derived from the Latin roots.
- Example, the common name of methanal is formaldehyde.
- Common name of substituted aldehydes can be given as follows,
- The common names of substituted aldehydes are named as derivatives of straight-chain parent compounds.
- Greek letters designate the carbon atoms near the carbonyl group.
- The carbon atom bonded to the carbonyl group is alpha, the next removed is beta and so on.

We use the suffix -carbaldehyde, when the carbonyl group \[\left( { - CHO} \right)\]is linked to the ring.
We can draw the structure of the given aldehyde as,

The parent carbon chain in the given compound is cyclopentane, the substituent is a hydroxyl group present at carbon in the second position. The carbonyl carbon will occupy the first position. Since the aldehyde group is attached to the cyclopentane ring, the name of the compound is 2-hydroxycyclopentane carbaldehyde.
So, the correct answer is “Option C”.
Note: We can prepare carboxylic acids by oxidizing aldehydes. We can also prepare alcohol by reducing aldehydes. Smaller aldehydes are sparingly water soluble, higher aldehydes are insoluble in water. Aldehydes are used in organic reactions such as,
- Woff-Kishner reduction
- Pinacol coupling reaction
- Oxo-Diels-Alder reaction
- Decarbonylation
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

Which chemical is known as king of chemicals A H 2 class 11 chemistry CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




