The correct experiment set-up for separating a substance by sublimation is:
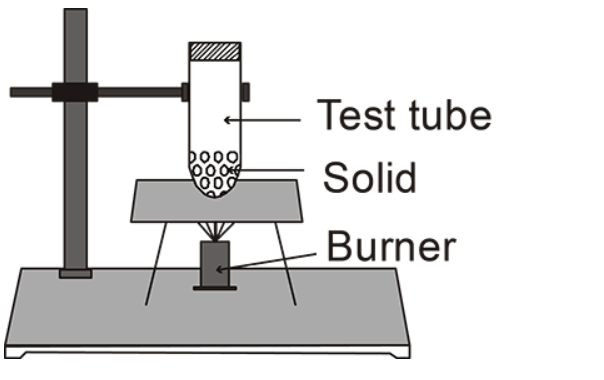
(A)
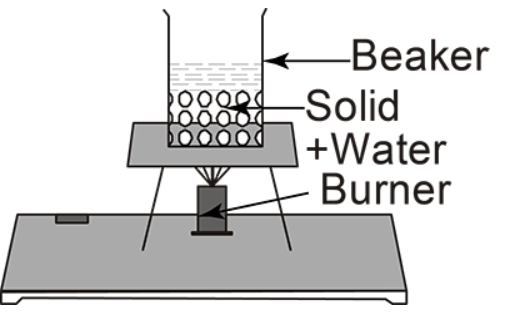
(B)
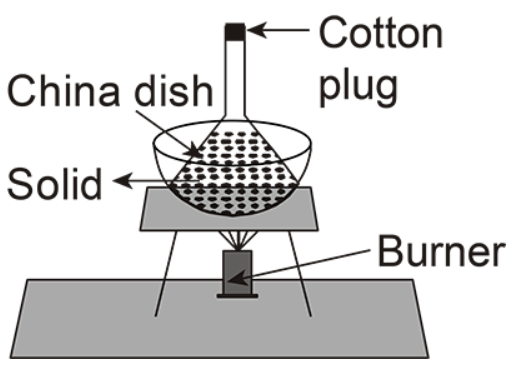
(C)
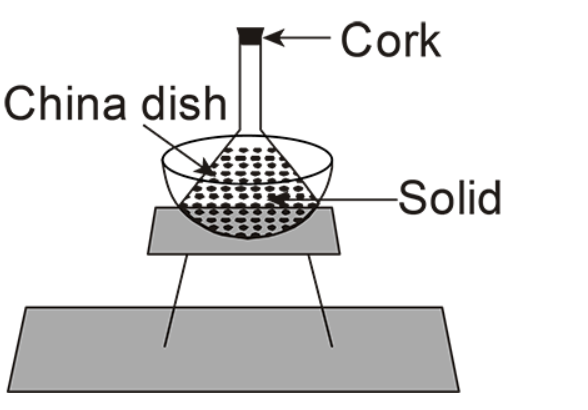
(D)




Answer
572.4k+ views
Hint :In the given options, C is the correct option.
So in order to separate a mixture from non-volatile compound to volatile one, sublimation process is used.
The mixture is first heated with the help of a burner. The solid compound sublimes after heating and turns into a gaseous state. Then the gas passes out from the cotton plug.
Complete Step By Step Answer:
In the given options, C is the correct option.
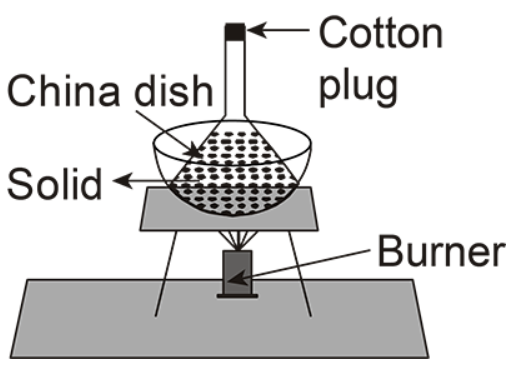
In this setup there is a base in which their burner is placed and a sheet is present at the base of the china dish, the solid is placed inside the funnel and the cotton plug is placed at the top of it.
Here, sublimation is generally a process in which the solid state phase is changed into gaseous state without converting it into a liquid form.
The mixture is first heated with the help of a burner. The solid compound sublimes after heating and turns into a gaseous state. Then the gas passes out from the cotton plug.
Note :
Sublimation is a process in which matter undergoes direct phase transition from a solid state to gas. This won’t involve the liquid transition state. For example we know that ammonium chloride is the perfect example of changing a solid to directly in tis gaseous form on constant heating. So in order to separate a mixture from non-volatile compound to volatile one, sublimation process is used.
So in order to separate a mixture from non-volatile compound to volatile one, sublimation process is used.
The mixture is first heated with the help of a burner. The solid compound sublimes after heating and turns into a gaseous state. Then the gas passes out from the cotton plug.
Complete Step By Step Answer:
In the given options, C is the correct option.

In this setup there is a base in which their burner is placed and a sheet is present at the base of the china dish, the solid is placed inside the funnel and the cotton plug is placed at the top of it.
Here, sublimation is generally a process in which the solid state phase is changed into gaseous state without converting it into a liquid form.
The mixture is first heated with the help of a burner. The solid compound sublimes after heating and turns into a gaseous state. Then the gas passes out from the cotton plug.
Note :
Sublimation is a process in which matter undergoes direct phase transition from a solid state to gas. This won’t involve the liquid transition state. For example we know that ammonium chloride is the perfect example of changing a solid to directly in tis gaseous form on constant heating. So in order to separate a mixture from non-volatile compound to volatile one, sublimation process is used.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Class 11 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




