The chemical formula of X, Y and Z are:
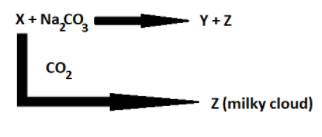
A. $CaO,Ca{\left( {OH} \right)_2},NaOH$
B. $NaOH,CaO,CaC{O_3}$
C. $NaOH,Ca{\left( {OH} \right)_2},CaC{O_3}$
D. $Ca{\left( {OH} \right)_2},NaOH,CaC{O_3}$
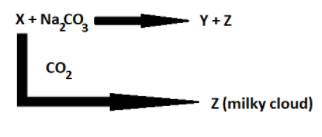
Answer
598.2k+ views
Hint: As the options are given in terms of Calcium, Find the compound of calcium which on reacting with Carbon dioxide forms a milky cloud. And then find the milky cloud compound and the products when calcium compound (X) is reacted with Sodium carbonate.
Complete step by step answer:
We are given two equations with unknown compounds and with clues. We have to find the compound names.
The only calcium compound when it is reacted with carbon dioxide turns into a milky cloud is Calcium hydroxide.
Calcium hydroxide is also known as slaked lime or lime water.
Calcium hydroxide reacts with carbon dioxide to form a white precipitate of calcium carbonate and water.
$Ca{\left( {OH} \right)_2} + C{O_2}\xrightarrow[{ - {H_2}O}]{}CaC{O_3}$
The milky cloud is the precipitate of calcium carbonate. Calcium carbonate is also known as Lime stone.
Therefore, X is Calcium hydroxide $Ca{\left( {OH} \right)_2}$ and Z is calcium carbonate $CaC{O_3}$
Now we have to find the compound Y.
X reacts with Sodium carbonate to give Y and Z, this means Calcium hydroxide reacts with Sodium carbonate to form Y and Calcium carbonate.
Writing the equation of reaction of Calcium hydroxide Sodium carbonate
$Ca{\left( {OH} \right)_2} + N{a_2}C{O_3} \to Y + CaC{O_3} \\
\therefore Y = 2NaOH \\
Ca{\left( {OH} \right)_2} + N{a_2}C{O_3} \to 2NaOH + CaC{O_3} \\$
Calcium hydroxide reacts with Sodium carbonate to form 2 moles of Sodium hydroxide and 1 mole of Calcium carbonate.
Therefore, the compound Y is NaOH.
X is $Ca{\left( {OH} \right)_2}$, Y is $NaOH$ and Z is $CaC{O_3}$.
The correct option is Option D, $Ca{\left( {OH} \right)_2},NaOH,CaC{O_3}$.
Note: Do not confuse sodium carbonate $N{a_2}C{O_3}$ with sodium bicarbonate $NaHC{O_3}$ and do not confuse calcium carbonate $CaC{O_3}$ with calcium bicarbonate $CaHC{O_3}$. The names and chemical formulas of these compounds are almost similar. So be careful when dealing with them.
Complete step by step answer:
We are given two equations with unknown compounds and with clues. We have to find the compound names.
The only calcium compound when it is reacted with carbon dioxide turns into a milky cloud is Calcium hydroxide.
Calcium hydroxide is also known as slaked lime or lime water.
Calcium hydroxide reacts with carbon dioxide to form a white precipitate of calcium carbonate and water.
$Ca{\left( {OH} \right)_2} + C{O_2}\xrightarrow[{ - {H_2}O}]{}CaC{O_3}$
The milky cloud is the precipitate of calcium carbonate. Calcium carbonate is also known as Lime stone.
Therefore, X is Calcium hydroxide $Ca{\left( {OH} \right)_2}$ and Z is calcium carbonate $CaC{O_3}$
Now we have to find the compound Y.
X reacts with Sodium carbonate to give Y and Z, this means Calcium hydroxide reacts with Sodium carbonate to form Y and Calcium carbonate.
Writing the equation of reaction of Calcium hydroxide Sodium carbonate
$Ca{\left( {OH} \right)_2} + N{a_2}C{O_3} \to Y + CaC{O_3} \\
\therefore Y = 2NaOH \\
Ca{\left( {OH} \right)_2} + N{a_2}C{O_3} \to 2NaOH + CaC{O_3} \\$
Calcium hydroxide reacts with Sodium carbonate to form 2 moles of Sodium hydroxide and 1 mole of Calcium carbonate.
Therefore, the compound Y is NaOH.
X is $Ca{\left( {OH} \right)_2}$, Y is $NaOH$ and Z is $CaC{O_3}$.
The correct option is Option D, $Ca{\left( {OH} \right)_2},NaOH,CaC{O_3}$.
Note: Do not confuse sodium carbonate $N{a_2}C{O_3}$ with sodium bicarbonate $NaHC{O_3}$ and do not confuse calcium carbonate $CaC{O_3}$ with calcium bicarbonate $CaHC{O_3}$. The names and chemical formulas of these compounds are almost similar. So be careful when dealing with them.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Which among the following are examples of coming together class 11 social science CBSE




