The anomeric carbon in D-(+)-glucose is:
A. C-1 carbon
B. C-2 carbon
C. C-5 carbon
D. C-6 carbon
Answer
607.2k+ views
Hint: Think about the definition of the anomeric carbon and how the placement of the hydroxyl group around it enables it to form the $\alpha $- and $\beta $- anomers of and given sugar.
Complete step by step solution:
- We know that anomeric carbon is carbon whose stereochemistry determines the type of anomer that the compound is. It is used to refer to only the cyclized forms of sugars.
- In the open chain form of the sugar, the anomeric carbon is known to be the carbonyl carbon that is involved in the aldehydic or the ketonic group. After cyclization, the anomeric carbon is found near the oxygen atom in the pyranose or furanose ring.
- Here, we are concerned about the pyranose form of glucose. First let us look at the Fischer projection formula and how it undergoes cyclization.
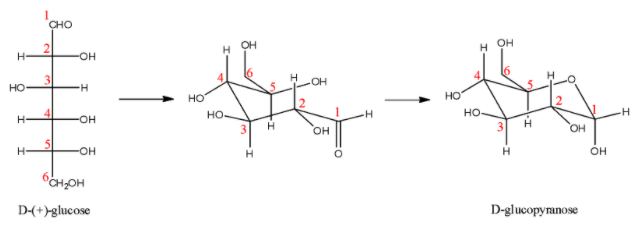
Here, we can see the carbon atom that is next to the oxygen atom in the pyranose ring, and we can determine that it is the same carbon that is present in the aldehydic group in the Fischer projection formula.
- The ring can close in two different ways: with the new OH group down or up in the conventional Haworth projection. This carbon atom which can assume either configuration is referred to as anomeric carbon and the two different cyclised forms as anomers of each other called $\alpha $- D – glucopyranose and $\beta $- D – glucopyranose. When the hydroxyl group on the C-1 atom is pointing downwards, it is called the $\alpha $- anomer and when it is pointing upwards, it is called the $\beta $- anomer. The structures are:
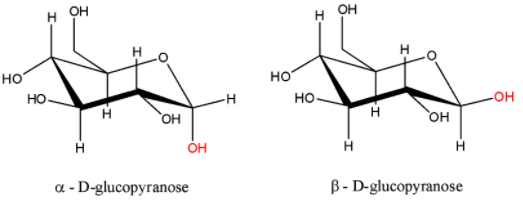
Therefore, from above we can easily conclude that option A is the correct option to the given question.
Note: An anomer is a special case of an epimer. An epimer is an isomer where the stereochemistry of a sugar is changed at one and only one chiral center. In anomers, that chiral centre happens to be the C-1 atom. D-glucose and D-galactose are epimers of each other.
Complete step by step solution:
- We know that anomeric carbon is carbon whose stereochemistry determines the type of anomer that the compound is. It is used to refer to only the cyclized forms of sugars.
- In the open chain form of the sugar, the anomeric carbon is known to be the carbonyl carbon that is involved in the aldehydic or the ketonic group. After cyclization, the anomeric carbon is found near the oxygen atom in the pyranose or furanose ring.
- Here, we are concerned about the pyranose form of glucose. First let us look at the Fischer projection formula and how it undergoes cyclization.

Here, we can see the carbon atom that is next to the oxygen atom in the pyranose ring, and we can determine that it is the same carbon that is present in the aldehydic group in the Fischer projection formula.
- The ring can close in two different ways: with the new OH group down or up in the conventional Haworth projection. This carbon atom which can assume either configuration is referred to as anomeric carbon and the two different cyclised forms as anomers of each other called $\alpha $- D – glucopyranose and $\beta $- D – glucopyranose. When the hydroxyl group on the C-1 atom is pointing downwards, it is called the $\alpha $- anomer and when it is pointing upwards, it is called the $\beta $- anomer. The structures are:

Therefore, from above we can easily conclude that option A is the correct option to the given question.
Note: An anomer is a special case of an epimer. An epimer is an isomer where the stereochemistry of a sugar is changed at one and only one chiral center. In anomers, that chiral centre happens to be the C-1 atom. D-glucose and D-galactose are epimers of each other.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE




