Study the roadmap for preparation of washing soda and fill up the blanks:
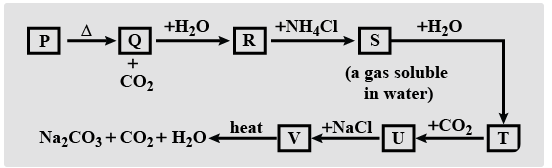
A. $\text{P-CaC}{{\text{O}}_{\text{3}}}\text{, Q-Cao, R-Ca(OH}{{\text{)}}_{\text{2}}}\text{, S-N}{{\text{H}}_{\text{3}}}\text{, T-N}{{\text{H}}_{\text{4}}}\text{OH, U-N}{{\text{H}}_{\text{4}}}\text{C}{{\text{O}}_{\text{3}}}\text{, V-NaHC}{{\text{O}}_{\text{3}}}$
B. \[\text{P-CaC}{{\text{l}}_{2}}\text{, Q-Cao, R-Ca(OH}{{\text{)}}_{\text{2}}}\text{, S-HCl, T-HCl, U-NaHC}{{\text{O}}_{3}}\text{, V-HCl}\]
C. \[\text{P-CaC}{{\text{l}}_{2}}\text{, Q-Cao, R-CaC}{{\text{O}}_{3}}\text{, S-N}{{\text{H}}_{3}}\text{, T-HCl, U-N}{{\text{H}}_{4}}\text{Cl, V-NaHC}{{\text{O}}_{3}}\]
D. \[\text{P-CaC}{{\text{O}}_{3}}\text{, Q-Cao, R-Ca(OH}{{\text{)}}_{2}}\text{, S-HCl, T-C}{{\text{l}}_{2}}\text{, U-CaC}{{\text{l}}_{2}}\text{, V-NaHC}{{\text{O}}_{3}}\]

Answer
621.9k+ views
Hint: Washing soda is a sodium carbonate which has 10 molecules of water of crystallization. It is also called sodium carbonate decahydrate.
Complete step by step answer:
- The formula of washing soda is $\text{N}{{\text{a}}_{\text{2}}}\text{C}{{\text{O}}_{\text{3}}}\text{.10}{{\text{H}}_{\text{2}}}\text{O}$ so our main purpose is to get sodium carbonate. Now let us see how we will do this.
- Take some amount of calcium carbonate (\[\text{CaC}{{\text{O}}_{\text{3}}}\]) heat it to give calcium oxide($\text{CaO}$) and carbon dioxide (as residual).
- Now react this calcium oxide ($\text{CaO}$) with water to give calcium hydroxide ($\text{Ca(OH}{{\text{)}}_{\text{2}}}$).
- Calcium hydroxide ($\text{Ca(OH}{{\text{)}}_{\text{2}}}$) in the presence of ammonium chloride($\text{N}{{\text{H}}_{\text{4}}}\text{Cl}$) will give us ammonia($\text{N}{{\text{H}}_{\text{3}}}$).
- The ammonia obtain in the last step reacts with water to give us ammonium hydroxide$\text{N}{{\text{H}}_{\text{4}}}\text{OH}$.
- This ammonium hydroxide in the presence of carbon dioxide changes into ammonium bicarbonate ($\text{N}{{\text{H}}_{\text{4}}}\text{HC}{{\text{O}}_{\text{3}}}$).
- This ammonium bicarbonate reacts with sodium chloride to give sodium bicarbonate hydrogen ($\text{NaHC}{{\text{O}}_{\text{3}}}$).
- This sodium bicarbonate is heated to give sodium carbonate ($\text{N}{{\text{a}}_{\text{2}}}\text{C}{{\text{O}}_{\text{3}}}$).
- This sodium bicarbonate is recrystallized to give washing soda ($\text{N}{{\text{a}}_{\text{2}}}\text{C}{{\text{O}}_{\text{3}}}\text{.10}{{\text{H}}_{\text{2}}}\text{O}$).
So, we can conclude that option A is correct.
Note: Whenever sodium carbonate crystals are exposed to air efflorescence takes place. Efflorescence is the phenomenon by which sodium carbonate losses water of crystallization and turns into white powder when exposed to air.
Complete step by step answer:
- The formula of washing soda is $\text{N}{{\text{a}}_{\text{2}}}\text{C}{{\text{O}}_{\text{3}}}\text{.10}{{\text{H}}_{\text{2}}}\text{O}$ so our main purpose is to get sodium carbonate. Now let us see how we will do this.
- Take some amount of calcium carbonate (\[\text{CaC}{{\text{O}}_{\text{3}}}\]) heat it to give calcium oxide($\text{CaO}$) and carbon dioxide (as residual).
- Now react this calcium oxide ($\text{CaO}$) with water to give calcium hydroxide ($\text{Ca(OH}{{\text{)}}_{\text{2}}}$).
- Calcium hydroxide ($\text{Ca(OH}{{\text{)}}_{\text{2}}}$) in the presence of ammonium chloride($\text{N}{{\text{H}}_{\text{4}}}\text{Cl}$) will give us ammonia($\text{N}{{\text{H}}_{\text{3}}}$).
- The ammonia obtain in the last step reacts with water to give us ammonium hydroxide$\text{N}{{\text{H}}_{\text{4}}}\text{OH}$.
- This ammonium hydroxide in the presence of carbon dioxide changes into ammonium bicarbonate ($\text{N}{{\text{H}}_{\text{4}}}\text{HC}{{\text{O}}_{\text{3}}}$).
- This ammonium bicarbonate reacts with sodium chloride to give sodium bicarbonate hydrogen ($\text{NaHC}{{\text{O}}_{\text{3}}}$).
- This sodium bicarbonate is heated to give sodium carbonate ($\text{N}{{\text{a}}_{\text{2}}}\text{C}{{\text{O}}_{\text{3}}}$).
- This sodium bicarbonate is recrystallized to give washing soda ($\text{N}{{\text{a}}_{\text{2}}}\text{C}{{\text{O}}_{\text{3}}}\text{.10}{{\text{H}}_{\text{2}}}\text{O}$).
So, we can conclude that option A is correct.
Note: Whenever sodium carbonate crystals are exposed to air efflorescence takes place. Efflorescence is the phenomenon by which sodium carbonate losses water of crystallization and turns into white powder when exposed to air.
Recently Updated Pages
Master Class 10 Computer Science: Engaging Questions & Answers for Success

Master Class 10 General Knowledge: Engaging Questions & Answers for Success

Master Class 10 English: Engaging Questions & Answers for Success

Master Class 10 Social Science: Engaging Questions & Answers for Success

Master Class 10 Maths: Engaging Questions & Answers for Success

Master Class 10 Science: Engaging Questions & Answers for Success

Trending doubts
Explain the Treaty of Vienna of 1815 class 10 social science CBSE

What is the median of the first 10 natural numbers class 10 maths CBSE

Who Won 36 Oscar Awards? Record Holder Revealed

What is the name of Japan Parliament?

Why is it 530 pm in india when it is 1200 afternoon class 10 social science CBSE

Select the word that is correctly spelled a Twelveth class 10 english CBSE




