How many structural isomers can be drawn for ${{\text{C}}_5}{{\text{H}}_{10}}$
Answer
585.3k+ views
Hint: The molecules having the same molecular formula but a different arrangement of atoms are known as isomers. First, we will determine the degree of unsaturation, so we will know the number of unsaturated bonds. Then we will draw the possible geometries.
Complete answer:
First we will calculate the degree of unsaturation as follows:
${\text{D}}{\text{.U}}\,{\text{ = }}\,{\text{C}} + 1\, - \frac{{{\text{no}}{\text{. of monovalent}}\, - \,{\text{no}}{\text{. of}}\,{\text{trivalent}}}}{2}$
On substituting $5$ for C and $10$ for no. of monovalent,
${\text{D}}{\text{.U}}\,{\text{ = }}\,5 + 1\, - \frac{{10}}{2}$
\[{\text{D}}{\text{.U}}\,{\text{ = }}\,5\, + 1 - 5\]
\[{\text{D}}{\text{.U}}\,{\text{ = 1}}\]
So, the degree of unsaturation is \[{\text{1}}\]. So, we have the possibility of the presence of one double bond.
The isomers having same molecular formula but different structures are known as structural isomers.
We can draw the five carbon atoms chain having double bond at first carbon as follows:
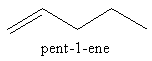
We can change the position of double bond to get a new structural isomer as follows:
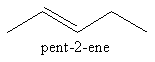
Now we can draw a four carbon chain having one methyl group as follows:
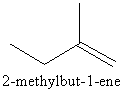
We can shift the position of methyl group from second carbon to third carbon as follows:
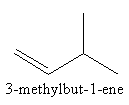
We can change the position of double bond from first carbon to second carbon in four carbon chain as follows:
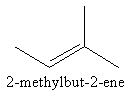
The above all structures have the same molecular formula but are different molecules.So, we can draw $5$ structural isomers of ${{\text{C}}_5}{{\text{H}}_{10}}$.
Therefore, the total number of structural isomers can be drawn for ${{\text{C}}_5}{{\text{H}}_{10}}$ is, $5$.
Note: Isomers have the same molecular formula but different chemical formula. In structural terms the connectivity differs. A molecular formula shows the total number of an atom in the compound. The chemical formula shows the different group of atoms of a molecule. Here, ${{\text{C}}_5}{{\text{H}}_{10}}$is the molecular formula but the ${\text{C}}{{\text{H}}_2}{\text{CHC}}{{\text{H}}_2}{\text{C}}{{\text{H}}_{\text{2}}}{\text{C}}{{\text{H}}_3}$ is the chemical formula of pentene. By the general formula of hydrocarbon, we will decide that the given molecular formula is representing the alkane, alkene, or alkyne. The general formula of the alkene is, ${{\text{C}}_{\text{n}}}{{\text{H}}_{{\text{2n}}}}$ .Where, n is the number of carbon atoms. So, ${{\text{C}}_5}{{\text{H}}_{10}}$is an alkene.
Complete answer:
First we will calculate the degree of unsaturation as follows:
${\text{D}}{\text{.U}}\,{\text{ = }}\,{\text{C}} + 1\, - \frac{{{\text{no}}{\text{. of monovalent}}\, - \,{\text{no}}{\text{. of}}\,{\text{trivalent}}}}{2}$
On substituting $5$ for C and $10$ for no. of monovalent,
${\text{D}}{\text{.U}}\,{\text{ = }}\,5 + 1\, - \frac{{10}}{2}$
\[{\text{D}}{\text{.U}}\,{\text{ = }}\,5\, + 1 - 5\]
\[{\text{D}}{\text{.U}}\,{\text{ = 1}}\]
So, the degree of unsaturation is \[{\text{1}}\]. So, we have the possibility of the presence of one double bond.
The isomers having same molecular formula but different structures are known as structural isomers.
We can draw the five carbon atoms chain having double bond at first carbon as follows:
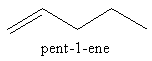
We can change the position of double bond to get a new structural isomer as follows:
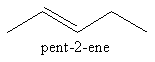
Now we can draw a four carbon chain having one methyl group as follows:

We can shift the position of methyl group from second carbon to third carbon as follows:

We can change the position of double bond from first carbon to second carbon in four carbon chain as follows:
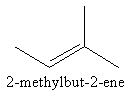
The above all structures have the same molecular formula but are different molecules.So, we can draw $5$ structural isomers of ${{\text{C}}_5}{{\text{H}}_{10}}$.
Therefore, the total number of structural isomers can be drawn for ${{\text{C}}_5}{{\text{H}}_{10}}$ is, $5$.
Note: Isomers have the same molecular formula but different chemical formula. In structural terms the connectivity differs. A molecular formula shows the total number of an atom in the compound. The chemical formula shows the different group of atoms of a molecule. Here, ${{\text{C}}_5}{{\text{H}}_{10}}$is the molecular formula but the ${\text{C}}{{\text{H}}_2}{\text{CHC}}{{\text{H}}_2}{\text{C}}{{\text{H}}_{\text{2}}}{\text{C}}{{\text{H}}_3}$ is the chemical formula of pentene. By the general formula of hydrocarbon, we will decide that the given molecular formula is representing the alkane, alkene, or alkyne. The general formula of the alkene is, ${{\text{C}}_{\text{n}}}{{\text{H}}_{{\text{2n}}}}$ .Where, n is the number of carbon atoms. So, ${{\text{C}}_5}{{\text{H}}_{10}}$is an alkene.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

How many of the following diseases can be studied with class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

10 examples of friction in our daily life




