
Statement A: Polyvinyl alcohol is obtained by polymerization of vinyl alcohol.
Statement R: Polyvinyl alcohol is prepared by hydrolysis of polyvinyl acetate.
(A) Both A and R are correct and R is the correct explanation of A.
(B) Both A and R are correct but R is not the correct explanation of A.
(C) A is correct but R is incorrect.
(D) A is incorrect but R is correct.
Answer
518.4k+ views
Hint :Polyvinyl alcohol (PVA) cannot be prepared by direct polymerization of vinyl alcohol. The synthesis is performed by the suspension polymerization of vinyl acetate. PVA or the polyvinyl alcohol is a widely used coating industry polymer, which is commonly used as textile fiber coatings or as coatings for the pills.
Complete Step By Step Answer:
PVA is a suitable polymer for biomedical applications due to its chemical properties and structure. The hydroxyl groups present make it soluble in water and in the body fluids.
Synthesis of PVA is accomplished by several methods, one of the simplest and the easiest to do is by the polymerization of vinyl alcohol.
But this cannot be done directly or there is a problem with this approach because vinyl alcohol is a very unstable chemical substance.
Vinyl alcohol is very easily tautomerized into acetaldehyde and acetaldehyde is a very stable resonance structure of vinyl alcohol. Because of this we need to apply a different approach towards the PVA.
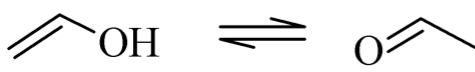
This method is the hydrolysis of polyvinyl acetate(PVAc)
This hydrolysis of PVAc into PVA implies the chemical modification where the acetate group leads to the alcohol group.
Hydrolyzing PVAc by methanolysis is conventional.
In this method the PVAc is dissolved in the methanol and after that adding a base or an acid in order to catalyse the solution.
And thereafter the acetate is hydrolyzed into the hydroxyl groups.
Here we need to adjust the time, temperature, and pH of the methanolysis.
Therefore from the above we can conclude that both the statements A and R are correct but R is not the correct explanation of A.
Note :
Degree of hydrolysis determines many mechanical properties that PVA possesses. By the adjustment of pH strength of the methanolysis different degrees of hydrolysis can be obtained.
Complete Step By Step Answer:
PVA is a suitable polymer for biomedical applications due to its chemical properties and structure. The hydroxyl groups present make it soluble in water and in the body fluids.
Synthesis of PVA is accomplished by several methods, one of the simplest and the easiest to do is by the polymerization of vinyl alcohol.
But this cannot be done directly or there is a problem with this approach because vinyl alcohol is a very unstable chemical substance.
Vinyl alcohol is very easily tautomerized into acetaldehyde and acetaldehyde is a very stable resonance structure of vinyl alcohol. Because of this we need to apply a different approach towards the PVA.
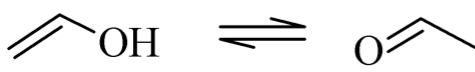
This method is the hydrolysis of polyvinyl acetate(PVAc)
This hydrolysis of PVAc into PVA implies the chemical modification where the acetate group leads to the alcohol group.
Hydrolyzing PVAc by methanolysis is conventional.
In this method the PVAc is dissolved in the methanol and after that adding a base or an acid in order to catalyse the solution.
And thereafter the acetate is hydrolyzed into the hydroxyl groups.
Here we need to adjust the time, temperature, and pH of the methanolysis.
Therefore from the above we can conclude that both the statements A and R are correct but R is not the correct explanation of A.
Note :
Degree of hydrolysis determines many mechanical properties that PVA possesses. By the adjustment of pH strength of the methanolysis different degrees of hydrolysis can be obtained.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




