What sort of hybridization does the central atom in \[S{F_4}\] have?
Answer
506.4k+ views
Hint: Atoms, the smallest unit into which matter is often divided without the discharge of electrically charged particles. It is also the smallest unit of matter that has the characteristic properties of an element. As such, the atom is the basic building block of chemistry.
Complete Answer:
The molecule will have a complement of \[34\] valence electrons, \[6\] from sulphur, and seven from each of the four fluorine atoms. In Sulphur tetrafluoride, the central atom of \[S{F_4}\] is \[s{p^3}d\] hybridized. Sulphur forms \[4\] single bonds and has \[1\] lone pair, which suggests that its steric number, which is that the name given to the number of regions of electron density, is adequate to \[5\] .
The \[S{F_4}\] Lewis structure is the combination of \[34\] electron and \[5\] electron pairs around the Sulphur, where there are \[4\] bonding pairs and \[1\] lone pair. This electron arrangement is named 'Trigonal Bipyramidal'. The rationale behind this is often that the lone pair prefers one among the equatorial positions.
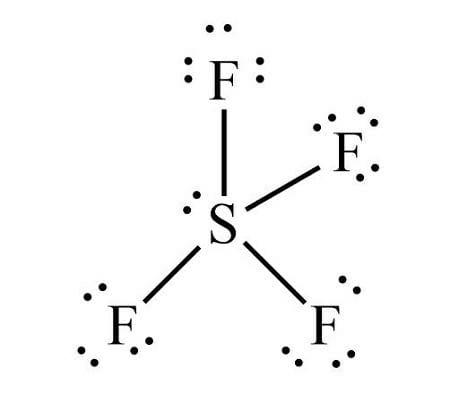
The example of the see-saw shape of the molecule is the sulfur tetrafluoride, or \[S{F_4}\] . Sulfur is the central atom, two fluorine atoms are on the equatorial plane, and two are on the axial plane.
Note:
Trigonal bipyramidal \[\left( {s{p^3}d} \right)\] is the shape of \[S{F_4}\] with one equatorial position occupied by \[1\] lone pair. It's a see-saw shape because it contains four bond pairs and one lone pair. The equatorial \[F\] atoms are \[120\] from one another, therefore the axial or equatorial bond angle is \[{90^ \circ }\] .
Complete Answer:
The molecule will have a complement of \[34\] valence electrons, \[6\] from sulphur, and seven from each of the four fluorine atoms. In Sulphur tetrafluoride, the central atom of \[S{F_4}\] is \[s{p^3}d\] hybridized. Sulphur forms \[4\] single bonds and has \[1\] lone pair, which suggests that its steric number, which is that the name given to the number of regions of electron density, is adequate to \[5\] .
The \[S{F_4}\] Lewis structure is the combination of \[34\] electron and \[5\] electron pairs around the Sulphur, where there are \[4\] bonding pairs and \[1\] lone pair. This electron arrangement is named 'Trigonal Bipyramidal'. The rationale behind this is often that the lone pair prefers one among the equatorial positions.

The example of the see-saw shape of the molecule is the sulfur tetrafluoride, or \[S{F_4}\] . Sulfur is the central atom, two fluorine atoms are on the equatorial plane, and two are on the axial plane.
Note:
Trigonal bipyramidal \[\left( {s{p^3}d} \right)\] is the shape of \[S{F_4}\] with one equatorial position occupied by \[1\] lone pair. It's a see-saw shape because it contains four bond pairs and one lone pair. The equatorial \[F\] atoms are \[120\] from one another, therefore the axial or equatorial bond angle is \[{90^ \circ }\] .
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

How many of the following diseases can be studied with class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which of the following enzymes is used for carboxylation class 11 biology CBSE

Explain zero factorial class 11 maths CBSE




